Process Puzzler: Vanquish Valve Vexations
This Month’s Puzzler
We’re having trouble with a reactor addition valve (see figure). It handles a minor ingredient that we add occasionally for a new product. We’re hoping to expand our business — and the valve is critical to that expansion. However, the addition valve is giving us trouble. I want to sort the problem out before the new product becomes a major part of our business.
We see traces of the new ingredient in quality testing. We see traces of caustic soda in the product, too.
I’m suspicious of the valve tee arrangement and the valve actuator performance. In addition, I want to pin down the source of the caustic soda. The main line is seldom down but the piping for the new ingredient gets periodically flushed and cleaned using a protocol of water flush, caustic wash and then water flush again. The flush return is upstream of a 2-in.-to-1-in. reducer.
What should I do to eliminate this contamination problem?
Scrutinize The Actuators
Start by taking a hard look at the valve and actuator specifications for the two valves. Actuators often are left to the vendor or project instrument engineer to specify. They’re actually (pun intended) pretty complicated devices. There are three types of actuators: air-to-close, spring-open; air-to-open, spring-close; and no spring, fail-last position. It sounds like the ingredient valve could be air-to-close, which is a bad choice because a bad instrument air supply could cause the spring to allow the valve to open, especially if there is a lot of back-pressure against the air diaphragm inside the actuator. If you find out the actuator was specified wrongly, replace it with one that has a higher inlet pressure and change the actuator to air-to-open; often this only requires a re-orientation of the actuator, as is the case for some clam-shell types.
There’s another force at play: the Venturi effect. A high-pressure flow can induce flow by creating a vacuum; this is how airplanes fly. The Venturi effect usually is a weak factor but keep in mind it’s there.
It’s also worth noting that the line necks down to 1 in. You’d get a tighter shutoff with a smaller valve. Then, it might be economical to consider an electrically actuated one.
As for the contamination, try to avoid cleaning one part of the pipe network individually. If you can’t avoid this, then install temporary blinds and make sure to drain to gravity. Otherwise, you risk bug growth from drains and contamination.
Another factor affecting cleaning and flow is the high viscosity. It may not seem like it but >50 cP is a high viscosity. Although some ordinary centrifugal pumps can function up to 300 cP, their hydraulic efficiencies are so low they may dead-head and require a prodigious amount of power. Honey is about 2,000 cP. Most liquid detergents are in the range of the new ingredient, about 100 cP. Given its high viscosity, getting all of the ingredient out of the pipe may be almost impossible. You may have to resort to far-more-expensive clean-out-of-place regimens instead of clean-in-place.
This should give you pause about the product expansion. If you can find an ingredient that is compatible with the new one, then consider pre-blending before pumping into the reactor.
Dirk Willard, consultant
Wooster, Ohio
August’s Puzzler
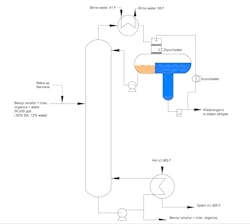
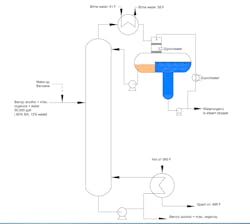
Our azeotropic distillation column serves as the first step in separating water from benzyl alcohol (Figure 1). Unfortunately, this trayed tower suffers periodic hiccups; flooding occurs, causing an off-specification bottom stream.
Initially, production tried boosting the reboiler temperature to reduce the periodic flooding. This led to condenser loading problems and high pressure in the condenser decanter that raises concerns about popping our pressure relief valve.
We looked at the feedstock for water contaminations. One operator suggested that the decanter is too small.
Another brainstorming session produced the idea of adding a feed preheater to vaporize the feed. The column designer says this is a bad idea. He warns it would increase the load on downstream columns because of more alcohol in the condensate. He suggests running a gamma back-scanner to determine where liquid is accumulating in the tower. His boss recommends a neutron back-scanner because of insulation.
What do you think is causing the problem? What tools should we use to determine the flooding tray(s)? What ideas do you have on how to address the periodic flooding?
Figure 1. Azeotropic distillation tower suffers sporadic flooding that causes an off-specification bottom stream.
Send us your comments, suggestions or solutions for this question by July 13, 2018. We’ll include as many of them as possible in the August 2018 issue and all on ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Rd., Suite 400N, Schaumburg, IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you’d like to pose to our readers, send it along and we’ll be pleased to consider it for publication.