Does A Double-Wall Tank Make Sense?
This Month’s Puzzler
Our sulfuric acid tank is corroded and leaking, a problem made even worse because our plant is located in a swamp next to wetlands of a major river. We’re desperate because the inspectors give us two years and we are squeezed against a creek. The only economically viable location is 100 ft from the creek. I am proposing a 12-ft diameter, 50,000-gal. double-walled storage tank without a diked-in containment area as the replacement. Corporate is concerned about not having a containment area. However, water bubbles out of the ground in our current diked areas. Moreover, a typical secondary containment area only lasts about 30 years before pipe suction lines buckle because the pumps are mounted on the area floors; we’ve tried re-lining dike floors without much success.
I’m proposing a simple pad for the pumps and heat exchangers, and another pad for the tank; the equipment pad will be connected to the tank pad by an expansion joint. Will this pass the regulators? Is a double-walled tank a better solution than a single-walled tank and secondary containment?
Ponder Three Points
In dealing with this problem, you should keep in mind potential liability resulting from a massive leak and disruptions in production, and assess three aspects.
Strategic considerations:
1. Valuable information could be had from tank manufacturers. You will need to develop appropriate specifications including, for example, the range of concentrations used, climate conditions, frequency of loading and unloading (as this will determine vent size) and routing of the vent (atmosphere, scrubber or other equipment).
2. Federal, state and local regulations — some with overlapping requirements — would apply for storage of hazardous materials. Keep relevant agencies posted on your work. Similarly, keep internal stakeholders in the loop.
3. Although Spill Prevention and Containment (SPCC) 40 CFR 112 does not apply directly to this storage, it has helpful guidelines for spill containment and management. Long-term, include periodic corrosion checks, leak detection and visual inspections.
Regulatory compliance and liability:
1. Because the site has had “bubbles from the ground,” conduct a soil study to determine its stability. You may need to stabilize the soil before installing the tank. For spill containment for large tanks (>10,000 gal), consider a lined-concrete dike with 110% capacity. Concrete reacts with the acid and, thus, will require a suitable and durable lining material. Double-wall construction, although a good containment safeguard, typically is used for smaller tanks. Loading/unloading stations also should include proper safeguards for spill containment/management. It’s preferable to keep pumps inside the dike to minimize penetrations of suction pipe through the concrete dike wall.
2. Proper material of construction is an effective safeguard against leaks. The concentration of sulfuric acid influences the material you should select for the tank and fittings. For example, carbon steel is a suitable metallurgy for 93% and above. Some vendors offer carbon steel for acid concentrations as low as 78% if the storage temperature is below 100°F. Non-metals such as high-density polyethylene are a good choice for applications within their size and temperature limitations, say, less than 10,000 gal and temperature not exceeding 120°F. Metallurgy considerations also apply to drains, vents and gaskets.
3. Sulfuric acid reacts with carbon steel; the products of the reaction are hydrogen and rust. Consider appropriate strainers for pumps. Keep the tank away from a source of spark or ignition. Molecular hydrogen can cause blistering, so periodic inspection is helpful.
4. Because sulfuric acid is highly hygroscopic and the water reaction is exothermic, avoid water ingress. Consider a nitrogen pad (split range control). Vent size should account for inflows and outflows and should suffice to handle all loading/unloading scenarios. Generally, vents are 3 in. or above. Personal exposure and safety considerations will dictate routing of the vents.
5. Ambient temperature variations matter. The freezing point of sulfuric acid is governed by its concentration. In cold climates, you may need to think about external insulation/heat tracing.
6. Consider external paint for protection from atmospheric corrosion.
Production:
1. One key objective is to maintain uninterrupted supply of sulfuric acid at the desired concentration range. Regular monitoring is vital for this.
2. Ensure systems are in place for detecting and rapidly responding to a spill.
GC Shah, Senior Advisor
Wood, Houston
Answer Some Key Questions
Economics and regulations will dictate the correct answer. There are lot of questions the company must answer. Once this is done, the path forward will be self-evident. Has the economics of each option been evaluated? Please address the following:
1. What are the regulations for tanks? The company must comply with and abide by the rules for personnel and neighborhood safety — and that includes the aquifer. Following these rules will be paramount.
2. If the regulations for tanks require dikes, then you must provide them. “No ifs or buts about it.” If a double-wall tank meets the regulations, you must compare its economics to that of a single-walled tank and secondary containment.
Girish Malhotra, president
EPCOT International, Pepper Pike, Ohio
Develop A Strong Case
In the 1990s, the EPA explored the use of double-walled tanks for just such a situation as you describe. Tanks were limited to about 12,000 gal. The idea turned out to be a raging success. Large tanks can be built.
I think your expectation for a containment area is a tad low. I’ve seen containment areas last more than 50 years.
Also, a 12-ft-dia tank requires a 60-ft height. I can see that you’re trying to keep the diameter below 14 ft so you can have the tank built in a shop. However, with that height, you must worry about wind loading and earthquakes. Besides, 60 ft is the limit of many lifts unless you have a crane handy. Instead, consider a 20-ft-dia tank that’s about 22-ft high. Unfortunately, it will require field-fabrication, which will cost about triple that of shop-building.
Now, let’s consider how to sell the idea of a double-walled tank.
My first suggestion is political: develop a complete FEL-2 design of both options: a single-walled tank with diked containment area and a double-walled tank with an equipment pad. Involve your environmental manager early because that person generally will know what regulators will accept. As a minimum, consider a sump with a pump to collect storm water. Add a conductivity alarm set for about double the total dissolved solids of the plant water. The environmental manager probably will want an additional level measurement. If so, opt for a submersible-head pressure transmitter; it can measure the static head of liquid in the sump, which can serve as a level input and a backup for the conductivity measurement. Include a French drain under the pad or the containment area with drainage to the sump. Put the engineering out for bid and then get bids on the resultant package from construction companies.
By doing both estimates, you can show corporate that doing it right — with a double-walled tank — costs the same or even less than doing it wrong with a single-walled tank. Certainly, the double-walled design will have a smaller footprint than a tank pad and containment dike. The EPA discourages dikes taller than 6 ft. So, for 50,000-gal storage, assuming a once-every-25-yr, 24-hr storm water intensity of 4-in/hr or 10% of the largest tank volume in the containment area (which gives a slightly lower storm-water accumulation), I reckon you will need 55 ft × 45-ft containment using a 4-ft dike. That’s a much bigger footprint than the 30-ft-dia tank pad and 20 ft × 15ft equipment pad needed by a double-walled tank. Note that a double-walled tank requires a minimum of 4 ft between the inner and outer walls and the pad requires an additional ft or so.
The cost of a double-walled tank likely will be about twice that of a single-walled tank. However, it offers significant immediate savings in civil work. Nevertheless, if you consider the time value of money, going with a single-walled tank still is a little less expensive. Unfortunately, many companies continue to choose the cheapest option, adding to the maintenance workload rather than putting problems behind them so they can work towards a better future.
Dirk Willard, consultant
Wooster, Ohio
July’s Puzzler
The filtration process for liquor leaving a treatment tank at my plant continues to plague us, despite a variety of changes made in the over a decade I’ve been there. It’s now of particular concern to me because I’ve worked my way up from operator to operations superintendent.
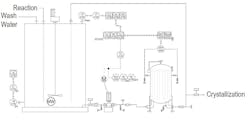
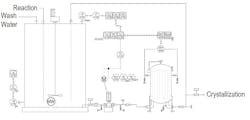
Figure 1. Liquor goes from a treatment tank to a sock filter before passing to recovery operations.
We started out with a crude filter but went to a cartridge filter, then to a filter press and now to a sock filter (see Figure 1). Corporate ditched the cartridge filter because the dirty cartridges added to our landfill, and soured on the press because it broke down a lot. A large horizontal pump provided the high pressure we required; that pump hardly ever fouled, although we deadheaded it a lot. We relocated the pH probe from the pump discharge to the treatment tank to ease access for maintenance. The old agitator in the tank was a 45° axial impeller but was replaced with a high-efficiency impeller to increase up/down flow. Corporate also swapped out the old level transmitter — a dP bubbler with a third tube to adjust for density, whose tubes typically fouled at the bottom well before our annual outage — with a radar gauge. It is great except when we steam the tank between batches to help clean the tank; then, we get a lot of high alarms.
The sock filter is a slight improvement over the filter press but takes up a huge amount of real estate. In addition, it poses some operational issues. We must use a lift to get the cages out of the housing; removing the solids is messy and a fire hazard. Corporate had promised that we wouldn’t have to change socks as often as we actually must and that it would be easier than cleaning the filter press. My quality engineer blames the problem on poor agitation in the tank.
How should I approach corporate to reduce the downtime and improve quality control in the process?
Send us your comments, suggestions or solutions for this question by June 12, 2020. We’ll include as many of them as possible in the July 2020 issue and all on ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Rd., Suite 400N, Schaumburg, IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you’d like to pose to our readers, send it along and we’ll be pleased to consider it for publication.