Keeping a chemical process running smoothly is one of the top priorities of plant management and operations. Process upsets and unplanned shutdowns interrupt production, lead to additional maintenance and extra costs, and generally cause headaches for everyone involved. As a result, a unit operation that experiences repeated upsets culminating in its shutdown and curtailed product output quickly becomes the focus of attention for plant personnel. This was the case in a cellulosics production unit at Dow's Midland, Mich., site — a steam stripper for removal of low levels of methanol and other lights from a process wastewater stream was suffering sudden unexplained pressure drop and level fluctuations that required shutdowns and restarts to resolve the tower's erratic performance.
Dow had selected steam stripping because it's a proven technology to remove methanol from wastewater, which reduces organic load on a treatment facility. The cellulosics production unit's tower has a rectifying section to concentrate the stripped methanol and other strippable organics, to lower the cost of incinerating the recovered methanol or possibly allow its use or sale as a byproduct. This turned the steam stripper into a full fractional distillation column.
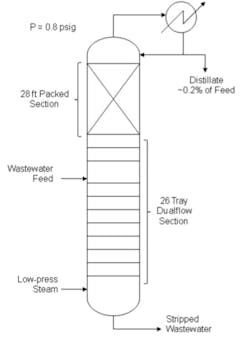
Wastewater distillation takes place in a 5-ft-diameter tower running slightly above atmospheric pressure with a total of 26 dualflow trays on a 24-in tray spacing and 28 ft of 40-mm ring-type random packing in a single bed (Figure 1). Twenty four of the dualflow trays are located below the wastewater feed point, with two trays immediately above the feed, and the packed bed above those two trays. Dualflow trays were selected for the stripping section for fouling resistance to potential solids and heavies in the wastewater feed, and the two trays above the feed were intended to handle any potential fouling due to entrainment from the feed section before the vapor enters the packed section. Heat comes from direct injection of 25-psig steam into the bottom of the tower to avoid any fouling issues with a reboiler.
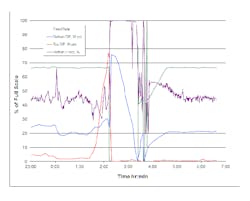
Figure 2. Steady pressure rise in packed section indicates most of the flooding occurs there.
A key tower design factor was the dilute nature of the contaminants in the wastewater feed stream. Methanol concentration averages about 2,000 ppm (mass) and ranges from 1,000 ppm to 2,500 ppm; the stream also includes about 200 ppm of other organics lighter than methanol and about 7,000 ppm of heavier non-strippable organics. Because the packed rectifying section would hold most of the water down in the tower, the overhead distillate would consist almost entirely of methanol and other strippable light components and its flow rate would amount to only a very small fraction of feed flow rate.
On the other hand, because methanol removal would require significant quantities of stripping steam, a substantial amount of the condensed overheads would go back into the top of the rectifying section as reflux. This would result in quite a high reflux ratio (reflux rate divided by distillate rate) and would require controlling the tower temperature profile via the low distillate rate rather than the more influential reflux rate. Moreover, regulating the tower this way would give a relatively slow control response because changes in the distillate rate would take time to affect the mass of liquid held up in the overheads system and the packed bed in the top of the tower.
ERRATIC PERFORMANCE
Upon startup of the tower in June 2005, the stripping section was able to remove >95% of the methanol in the wastewater feed with the design flow rate of stripping steam. However, the packed rectifying section was underperforming — far more water remained in the distillate, 3-5% (by mass), than the 0.1% or less expected from computer simulations. In addition, the tower experienced flooding in the top packed section at irregular but frequent intervals. Pressure drop across the packed section went as high as 8 psi (versus the <0.5 psi expected) before Operations was forced to shut down the unit due to degrading control of the tower levels and initiate a restart. Following the restart, the tower would settle back into stable operation for a time until the next flooding episode would occur anywhere from 12 hours to several days later.
Figure 2 shows tower response during a typical flooding episode. Note that pressure drop across the bottom trayed section remains fairly stable while the packed-section pressure drop steadily increases, indicating flooding is taking place mostly in the packed section. The sudden rise in trayed-section differential pressure after the cutoff of wastewater and steam feeds stems from the significant liquid holdup in the tower suddenly dropping into the bottom of the tower and filling it up above the bottom pressure transmitter tap used to calculate the trayed-section pressure drop.
A number of possibilities could explain the flooding: under-design of the tower, improper installation of tower internals, damage to the internals, fouling, and foaming. We verified design calculations and vapor/liquid equilibria. A full gamma scan of the tower three months after startup revealed no mechanical abnormalities, normal liquid holdups, and a possibility of only minor foaming on the trays just below the feed point. However, that scan took place when tower pressure drops were normal, not during a flooding episode. During a subsequent shutdown and opening of the tower, a vessel entry and visual inspection showed all tower internals to be in place, properly installed and undamaged with no sign of fouling of any kind.
Flooding episodes continued to occur. Then, a chemist who conducted periodic analyses of the distillate stream remarked that a partially obscured minor unknown peak in the distillate gas chromatograms frequently appeared. Further analytical work with mass spectrometry identified the unknown as 1,2-dimethoxypropane (DMOP), a component not shown in analyses of wastewater feed samples but one likely (from knowledge of the production process) to be present at low levels. It was hypothesized that the presence of DMOP in the wastewater feed and its possible buildup in the top of the tower could prompt the flooding episodes.
TESTING THE HYPOTHESIS
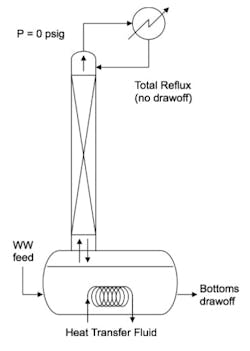
The lab performed continuous distillation on drum quantities of actual wastewater feed. A 2-in-diameter glass packed column was continuously bottom-fed with wastewater and operated at total reflux, as shown schematically in Figure 3.
With the continuous feed of wastewater to the bottom of the column with continuous drawoff of the bottoms to maintain a constant liquid level in the pot, the lab column configuration roughly matched the top portion of the production tower. In addition, the heat input to the pot was regulated so the reflux-to-feed rate ratio was the same as that in the production tower. Operating the lab column at total reflux enabled testing the potential for DMOP to accumulate in the top of the column because there would be no opportunity for purging any component from the top.
Results showed that with a low but unquantifiable level of DMOP in the wastewater feed, DMOP indeed built up to significant concentrations in the top of the column with a nondetectable level of DMOP in the bottom drawoff from the column. In addition, as the experiment progressed, the concentrations of DMOP and water in the top of the column steadily decreased as the concentration of methanol and other lights built up (Table 1).
The buildup and subsequent falloff of DMOP in the top of the lab column, along with the nondetectable bottom DMOP concentration, indicated that DMOP is easily stripped from water but is less volatile than methanol. This provided some evidence that with a significant stripping section below the feed point, the DMOP entering in the feed couldn't be purged out the bottom of the production tower. In addition, a high reflux ratio in the rectifying section would keep DMOP out of the distillate so that it indeed might be building up in the top portion of the production tower to cause flooding. Every time the tower had to be shut down after a flooding episode, its bottom had to be pumped out to get rid of liquid that had accumulated in the top and dropped into the bottom. This removed the accumulated DMOP.
Upon restarting, the tower worked fine until DMOP again built up enough to cause another flooding episode. Effectively, the DMOP was behaving as a "trapped" component in the distillation tower — insufficient DMOP was leaving in the combined top and bottom streams to compensate for the influx of fresh DMOP in the entering wastewater.
Subsequent vapor/liquid equilibrium measurements indicated DMOP and methanol don't form an azeotrope at atmospheric pressure, and the relative volatility of methanol to DMOP at atmospheric pressure and high concentrations of methanol is 1.4. This verified that DMOP is heavier than methanol in the top of the production tower. Furthermore, measurements showed the relative volatility of DMOP to water at atmospheric pressure and low concentrations of DMOP to be about 70–100, compared to a relative volatility of 8 for methanol at low concentrations in water. These data confirmed that in the production tower DMOP is well stripped out of the wastewater exiting the bottom but can be held down out of the distillate by the high reflux ratio and the tall bed of packing in the top of the tower and thus become a trapped component.
Before the lab distillation and vapor/liquid equilibrium experiments were completed, repeated flooding episodes in the production tower eventually caused damage to its internals, as indicated by very high water concentrations (about 30%) in the distillate and high pressure drop through the condenser. Another gamma scan of the tower revealed that roughly half of the packed bed was missing. So, the tower once again was shut down and reopened. A visual inspection showed that the bed limiter and liquid distributor were damaged and the missing packing was in the vapor line and condenser, which explained the high pressure drop through the condenser. However, the trays were undamaged and in good condition.
THE SOLUTION
In June 2006, after repair of the damaged internals and replacement of the packing, the tower was restarted and its operation was changed to combat buildup of DMOP. Previously, tower control involved manipulating the distillate rate to maintain a representative temperature point in the top section that gave a distillate containing 3–5% water. However, that low a concentration of water didn't allow enough DMOP to exit the top of the tower to prevent it from being trapped and causing flooding. So, the tower temperature set point was raised to allow 5–10% water in the distillate. At that water concentration, sufficient DMOP leaves in the distillate stream to prevent buildup. The flooding episodes ceased.
In conclusion, recovery of low levels of organics from process wastewater by stripping with rectification up to high organic concentrations can lead to unexpected results such as trapped components in a tower. This can occur with complex organic/water-vapor/liquid equilibrium relationships that cause significantly different distillation behavior between the distillate and bottoms. However, methodical investigation into the causes of intractable distillation problems with well-conceived laboratory experiments, thorough data analysis, and understanding of the underlying principles usually will yield viable solutions.
BRUCE S. HOLDEN is principal research scientist in the Engineering and Process Sciences group of The Dow Chemical Co., Midland, Mich., PATRICK H. AU-YEUNG is a research scientist, Engineering and Process Sciences, in Midland, and TODD W. KAJDAN is an associate analytical manager at the Analytical Technology Center in Midland. E-mail them at [email protected], [email protected] and [email protected].