Researchers at the University of Illinois, Champaign, Ill., and the University of Birmingham (UB), Birmingham, U.K., report they have discovered a cheaper, more environmentally friendly and renewable catalyst for processing biofuels. The method uses bacteria and palladium recovered from wastes such as discarded electronics.
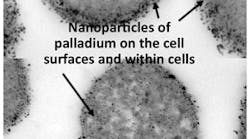
This photomicrograph shows the palladium and bacteria catalyst. Source: Lynne Macaskie.
Previous studies have shown that bio-oil flowing through porous carbon particles infused with palladium is effective. However, those carbon particles are not cheap, notes Illinois Sustainability Technology Center researcher B.K. Sharma, who led the research.
“Instead of using commercially produced carbon particles, we can use bacteria cell masses as a sort of biologic scaffolding for the palladium to hold onto,” Sharma explains. “The oil can flow through the palladium-decorated bacteria masses as it does through the carbon particles.”
“We found our product [called bio-Pd/C] to be as good or even slightly better [than commercially produced catalyst],” he notes. “We were able to remove the oxygen and nitrogen impurities at a comparable rate, and yielded the same volume of product using our cheaper, greener catalyst as is observed using the more expensive commercial catalyst,”
More details were published in the journal Fuel.
While the commercial catalyst can be used repeatedly without extensive processing, the bio-Pd/C must undergo processing to be reused.
“It is a minor caveat,” Sharma admits. “The fact that we have shown the potential of making refinery-ready crude oil from algae bio-oil using a catalyst that can be prepared from low-grade recycled metals and green and economical bacterial biomass proves that this is a very promising advancement. In addition, this bio-catalyst would work equally well in petrochemical processing.”
Lynne Macaskie of UB notes that with simple low-viscosity liquid streams the catalyst is easily immobilized and can be re-used multiple times whereas the commercial catalyst suffers attrition and loss.
“We are exploring how long we can use this catalyst without affecting performance and, also, how best we can re-utilize the metal from the spent catalyst for making the next batch,” adds Sharma.
“For bio-oil work, further optimization is needed to increase conversions to bio-oils from biomass and maximizing carbon utilization efficiency throughout the whole processing chain,” he says.
While scalability of catalyst production isn’t an issue, Macaskie explains that a proper lifecycle analysis is needed to identify bottlenecks and what can be done to overcome them. “This is in progress so that onward questions are fully formed and informed,” she adds.
If all goes as hoped, the researchers anticipate a developmental project over the next three years that will then dovetail into a suitable pilot test site.
“When the project started (looking at upgrading heavy oils from the Alberta Oilsands) we were granted access by the participating oil company to its pilot oil upgrading rig in Calgary. Further work on the oil would be in collaboration with a suitable pilot site and specialist chemical engineers. The source biomass for pyrolysis oil is algae, and a parallel line of work involves boosting biomass yield at the expense of sunlight, says Macaskie.
The team is collaborating with a company to make and test the catalyst for green chemistry applications in fine chemicals synthesis. They have another firm interested in making drop-in fuels from energy biomass byproducts.