Using a copper tetramer as catalyst could speed up conversion of carbon dioxide to methanol while ultimately saving energy, say researchers at the U.S. Department of Energy’s (DOE) Argonne National Laboratory, Lemont, Ill.
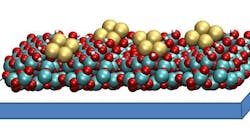
Figure 1. Catalyst consisting of small clusters of four copper atoms each, supported on a thin film of aluminum oxide, converts carbon dioxide more efficiently. Source: Larry Curtiss, Argonne National Laboratory.
The current process to reduce carbon dioxide to methanol uses a copper, zinc oxide and aluminum oxide catalyst and requires energy-intensive, high-pressure conditions to enable the atoms to bind to carbon dioxide.
In comparison, the copper tetratamer consists of small clusters of four copper atoms each, supported on a thin film of aluminum oxide (Figure 1). The catalyst attaches more strongly to carbon dioxide and can better accelerate the conversion, the researchers note. In addition, the process requires lower pressure and less energy to produce the same amount of methanol. More details appear in an article in a recent issue of the Journal of the American Chemical Society.
“With our catalyst, there is no inside,” explains Stefan Vajda, senior chemist at Argonne and the Institute for Molecular Engineering and coauthor on the paper. “All four copper atoms are participating because with only a few of them in the cluster, they are all exposed and able to bind.”
The catalyst can vary in size, composition and support material, creating tens of thousands of possible options; the researchers have narrowed down the possibilities to 2,000 combinations based on calculations factoring in the number of atoms in the cluster, the elements the cluster is made of, the support’s chemical composition and reaction conditions.
“We’re interested in finding new catalytic reactions that will be more efficient than the current catalysts, especially in terms of saving energy,” notes Larry Curtiss, an Argonne Distinguished Fellow and coauthor of the paper.
The team currently is pursuing tests on an expanded range of copper clusters sizes in combination with a variety of support materials.
“In our ongoing experimental and theoretical studies, we are focusing on copper clusters containing up to about 20 atoms and on the use of about a half-a-dozen support materials,” elaborates Vajda.
“We haven’t yet found a catalyst better than the copper tetramer, but we hope to,” he adds. “With global warming becoming a bigger burden, it’s pressing that we keep trying to turn carbon dioxide emissions back into something useful.”
Other challenges include instability and figuring out how to scale up the process. “There’s a chance that copper tetramers may decompose when put to use in an industrial setting, so ensuring long-term durability is a critical step for future research,” Curtiss notes.
“… There are several routes which offer viable options to stabilize the clusters. One approach is to use supports with suitable nanostructured morphologies and binding/defect sites for anchoring the clusters. Another approach is to “lock” the subnanometer particles inside (sub)nanometer size “craters” created by building walls made of oxide various oxide layers around the particles, or to pin the clusters into to support,” explains Vajda.
To scale up the process, “the fabrication of acceptably well-defined subnanometer-size particles (i.e., sufficiently narrow distributions and compositions) may need the development of new synthesis routes,” Vajda admits.
However, the researchers are strictly focused on identifying and understanding the properties of new materials. “Scaling up the production of the catalysts and a pilot-plant scale testing are out of the scope of our current studies, [but] Argonne has efforts on taking technologies closer to commercial scale, and the lab is always looking to improve our collaborations with industry,” he adds.
A company active in the development of renewable energy solutions has shown interest in further development of the technology.