Shape Selectivity Spurs Separation
Intrinsically porous organic cage molecules promise a more-energy-efficient option than distillation for separating C8 and C9 aromatic isomers and other molecules that differ mainly in their shape, report researchers at the University of Liverpool, U.K. Tailoring the shape-selective cages (Figure 1) for specific molecular separations is possible, they add. Moreover, the materials offer advantages over extended frameworks like inorganic zeolites and hybrid metal-organic frameworks (MOFs), the Liverpool team believes. (For details about a new "yoke-shell" design MOF, see "Porous Nanostructure Boosts Selectivity").
SEPARATION MECHANISM
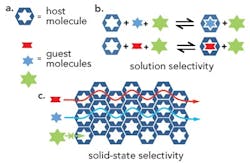
Figure 1. Shape selectivity of crystalline organic cage efficiently separates molecules. Source: University of Liverpool."We were able to demonstrate this new molecule separation technique by synthesizing porous organic cage molecules that are highly similar in shape to the molecules that need to be separated," explains Andrew Cooper, a professor in the university's department of chemistry and leader of the team. In laboratory trials, the shape sorting provided by such a crystalline cage molecule enabled separating a common aromatic feedstock, mesitylene, from its structural isomer, 4-ethyltoluene, with unprecedented perfect specificity. Moreover, unlike zeolites and MOFs, the cages are soluble in common organic solvents. This allows them to be readily processed into well-defined nanoparticles and co-processed directly into composite films and membranes, note the researchers. More details appear in a recent article in Nature Chemistry."We are looking at a range of separations, including hard-to-separate gas mixtures and chiral materials. As well as separating by shape, there is potential to separate by size, polarity, polarizability or chirality, or combinations of these properties," says Cooper. "The new work is already ongoing, and we would expect to have new separations in the next 12 months… We are attempting a scaled-up breakthrough measurement trial with partners in the U.S.A. in May.""We can prepare porous cage solids that are homochiral — hence, unlike porous materials derivatized with chiral functional groups, the entire solid is comprised of chiral entities. In principle, this might give big advantages for chiral separations. In terms of industrially relevant targets, we are interested in gas pairs such as ethane/ethene, propane/propene and krypton/xenon — all of these are very challenging separations," he adds. "We have just started work on chiral separations."The robustness of the organic cage depends upon the particular molecule. Some are quite robust and scale-up efforts are focusing on them, notes Cooper. The molecules likely are no better or worse than other microporous solids when it comes to susceptibility to clogging, he believes.Purely microporous materials can pose mass-transfer limitations in some applications, Cooper admits. However, the solution processability of the porous organic cage molecules offers an advantage in potential strategies to address such issues, he says.