Adsorption Gets A New Option
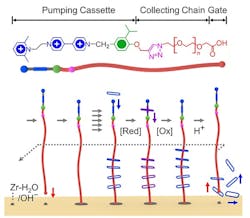
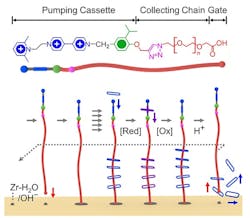
Figure 1. Redox method attracts a fleet of rings on and off the surface of a solid-state two-dimensional metal-organic framework. Source: Science.
Researchers at Northwestern University, Evanston, Ill, have developed what they term a fundamentally new mode of adsorption, called mechanisorption, that uses machine-like movements to attract molecules onto a solid surface. The researchers reckon the method holds broad promise, including for catalysts, energy storage and environmental remediation.
The research demonstrates how artificial molecular machines — synthetic molecular components that produce machine-like movements — grafted on surfaces can lure molecules at very high concentrations, thereby storing significant amounts of energy.
“If chemists can work out how mechanisorption can be incorporated in active structures, the storage of gases like hydrogen, carbon dioxide and methane will enter a whole new world and become a different ball game altogether,” believes Northwestern’s Fraser Stoddart, who received the 2016 Nobel Prize in Chemistry for his work involving the design and synthesis of molecular machines and led the research.
Mechanisorption results from non-equilibrium pumping cassettes forming mechanical bonds between the adsorbent (the surface) and the adsorbate (the molecules). The mechanism uses redox and acid-base chemistry to adsorb and desorb a fleet of rings onto and off the surface of a solid-state two-dimensional metal-organic framework.
Current work has focused on recruitment of ring molecules to surfaces, but the researchers anticipate these rings can be functionalized to bring many different types of molecules at high concentration to surfaces.
The researchers report in an article in the journal Science that the pumping efficiency decreases slightly with increasing number of redox cycles. However, recyclability is not a problem here, they say. “Regeneration — unloading the pumps and rings and the regrafting of the pumps — is a similar process as we regenerate adsorbents that do chemisorption, explains Liang Feng, who works in Stoddart’s lab.
The mechanisorption is highly selective, the researchers note, and only selective to the ring reported in the article. “Fundamentally, it can only uptake the rings we targeted so it can separate almost any other molecules from the rings. In the future, we will expand the diversity of the pumping cassettes to expand the separation scope,” comments Feng.
Potential industrial applications of the technology include catalysis, energy storage, supercapacitor, sequence-controlled synthesis of polymer, separation, adsorption, and electrochemistry. “Basically, everything related to surface chemistry. Our discovery is transformative in chemistry at surfaces and interfaces. Scaling-up is also possible without notable issues,” notes Feng.
The team currently is using mechanisorption to study the sequence-controlled synthesis of polymers, carbon capture, and many other applications, including catalysis and separation. “It might take 3–5 years,” says Feng.