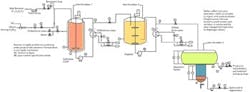
IMPROVED PROCESS
Figure 3. Addressing the problems in the sulfonation system requires a variety of modifications. Source: Dirk Willard.
If energy balance calculations show additional changes are needed, it may be necessary to add a smaller TV for split-range control of the reactor chiller temperature during the transition periods between steady operation and shutdown. A threat from your regeneration service company should not be taken lightly. One word from them and you're out of business. I see filters in your future. You can approach the spent-acid-contamination problem in two ways: a side-stream filtration for a modest problem or full-stream filtration. A hot water wash prior to filtration could help improve separation by avoiding gelling in the acid. Perform laboratory tests to identify the maximum pore size that will produce clear acid in combination with the optimum pre-treatment. Then, work backward to satisfy the service company, balancing economics and quality.Cleaning up the product may be easy once the source of trouble is identified. The entire process should be shut down and flushed of contaminants. Pay attention to the heat exchangers, especially as they may have significant fouling that has gone undetected. After the temperature control problem is ameliorated, the contaminant in the solid should go away. If not, you may want to take samples from the separator, adding filtration prior to neutralization when a slurry is produced, and a wash-centrifuge step prior to the spray dryer.There is no reasonable explanation for the corrosion failure of the Coriolis meter. Check valves leak, and if corroded badly, springs fail to close and acid flows back into the line supplying alkyl benzene. Install a backflow preventer to avoid liquid backflow of the H
2SO
4. It's doubtful that the acid vapor can be so easily contained. The only idea that comes to mind is a cold water wash followed by decanting; the acid vapor would go into the water but some trace alkyl benzene will be lost. You will want to chase the corrosion backward to the piston pump. These pumps are expensive to replace and long on delivery. In the end, it may be much easier to upgrade the pipe specification if the corrosion problem continues.
Dirk Willard, consultantWooster, OhioFEBRUARY'S PUZZLERMy refinery is grappling with how to set up the coolers for reducing oil sample temperatures for safe handling. These coolers usually consist of a U-tube inside a water jacket. The tubing is rated for 2,000 psig at 600°F; the shell maximum allowable working pressure is 75 psig at 600°F. We're working with heavy gas oil from a coker at 400°F. An alternative that's been suggested is to use a tube inside a tube: ¼-in. tubing for the oil within 1-in.tubing for the cooling-tower water. The project engineer wants hydrostatic pressure relief valves (PSVs) installed for blocked-in water. The maintenance engineer is pushing for the tube-in-tube approach because he insists it won't need a PSV; he claims it is protected under ASME code because the cooler diameter is less than 6-inch. Who's right? How should we proceed?Send us your comments, suggestions or solutions for this question by
January 10, 2014. We'll include as many of them as possible in the
February 2014 issue and all on
ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at
[email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Road, Suite 400N, Schaumburg IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.And, of course, if you have a process problem you'd like to pose to our readers, send it along and we'll be pleased to consider it for publication.