Metal-Free Catalyst Enhances Polymerization
A novel method for controlled cationic polymerization promises higher quality polymers, report researchers at the Nagoya Institute of Technology, Nagoya, Japan. The technique relies on weak halogen bonding and the addition of a small amount of ammonium salt to produce long homogeneous polymers. The researchers produced polyvinyl ethers of a predictable molecular weight and narrow molecular weight distribution using the method. Such metal-free polymers boast biocompatibility and have potential for biomaterials and semiconductors, the Japanese scientists believe.
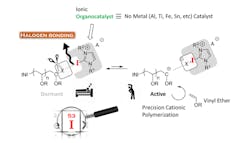
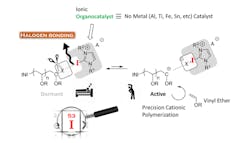
Figure 1. Method yields polyvinyl ether with predictable molecular weight and narrow molecular weight distribution. Source: Nagoya Institute of Technology.
“Metal halide catalysts… create impurities responsible for the coloration and degradation of polymers. There is a high demand for metal-free initiating systems,” says associate professor Koji Takagi, lead author of a recent paper in Chemistry – A European Journal that describes the new method. Producing a long homogeneous polymer without using metal-based catalysts is challenging, though, the researchers note.
Takagi and his coworkers developed a metal-free process for controlled cationic polymerization of isobutyl vinyl ether (IBVE) mediated by hydrogen bonding (Figure 1). It uses a halogen initiator together with an ionic organocatalyst. The catalyst abstracts from the initiator an anion that reversibly interacts with the catalyst and the propagating end of the polymer. Weak bonding is preferable to avoid side effects that might compromise the homogeneity and size of the resultant polymers. The researchers found that chloride forms appropriately weak halogen bonds with a 2-iodoimidazolium catalyst for polymerizing IBVE.
The chloride anion efficiently could shift back and forth between the catalyst and the propagating end of the IBVE, yielding a polymer with a high molecular weight and a narrow molecular weight distribution. Operating at a temperature of -10°C and with 10-mM concentrations for each element of the initiating system minimized deleterious side effects, say the researchers.
“Adding ammonium salt allowed us to modify the polymerization rate and molecular weight distribution,” explains Takagi.
The polymerization system is robust and boasts tunable catalytic activity as well as recyclability, among other advantages, he adds. The researchers now are turning to the application of the polymers prepared by the method.