Keep Under-Deposit Corrosion Under Control
Many plants are running at reduced rates at the moment. This raises a potential issue with piping. Economics usually result in fluid velocities in excess of 5 ft/sec for most systems. However, sustained operation at low rates due to COVID-19 may foster sedimentation at spots in piping systems where it never previously occurred — and create the potential for under-deposit corrosion (UDC). While the minimum velocity for sedimentation varies depending upon the system, any pipe with a liquid velocity under 1.5 ft/sec has a high possibility of sedimentation if solids are present.
[pullquote]
Even nominally 100% liquid systems may contain some solids. Such solids may come as feed contaminants, corrosion products, debris from reactors (catalysts) or driers, or low concentration products from normally ignored side reactions. Solids also may result from precipitation. At lower velocities, residence times rise; so, a heat loss or gain from the outside (either of which may produce solids) becomes more important.
A sedimented solid layer creates an area of the pipe under the solids whose chemistry differs from that of the area wetted by the liquid. First, the solid itself has a different composition. Second, the residence time of the liquid trapped inside the solid deposit is much longer. Reactions that have little significance in the bulk flow may have time to go to completion. In many circumstances, the deposit sets up an electrochemical cell that causes corrosion; this may happen in systems with either aqueous or organic flows.
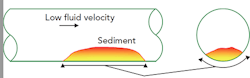
Let’s look at a deposit formed in one specific system (Figure 1). It handles a hydrocarbon fluid with ~0.1% free water. The velocity was low, so a solid deposit formed. This deposit consisted of iron oxide as well as 26.4 wt-% solid organics, 7.3 wt-% trapped moisture containing traces of oxygen, and ppm levels of chlorides and hydrogen sulfide.
Figure 1. Area under the deposit has different chemistry and no access to fresh liquid.
UDC generally includes multiple steps: deposit formation; onset of primary corrosion; oxygen depletion; start of secondary corrosion; and acceleration of acidic corrosion.
In steel piping systems, the primary corrosion mechanism after the deposit forms is: 2Fe + O2 + 2H2O → 2Fe2+ + 4OH- followed by 4Fe(OH)2 + O2 + xH2O → 2Fe2O3 + (x + 4)H2O. These reactions deplete free oxygen and continue as long as free oxygen and moisture are in contact at the pipe wall. While this is the most common UDC route, some systems don’t involve the oxygen depletion step.
The secondary corrosion occurs after the galvanic cell is set up. Finally, if the correct ions are present, the pH drops and the water trapped in the deposit becomes more acidic. The lower pH accelerates the corrosion rate.
UDC can pose a double whammy. It can lead to very aggressive (fast) localized corrosion, and can arise in dispersed places. The corrosion will occur where deposits appear. In this case, the deposits formed on the bottom of the pipe in the six-o’clock position. The corrosion was severe enough that it caused a hole all the way through the pipe — and liquid leaked into the insulation. A plant’s normal monitoring steps may not catch such trouble-spots if they arise in unusual locations.
So, low rate operations demand increased monitoring. The best approach for identifying locations requiring checking involves evaluating the process and mechanical layout, as well as conducting a historical review. (Heading off problems by adopting suitable preventive maintenance techniques also makes sense.)
Look at streams that potentially could form deposits from low velocity. Can low flow conditions allow sedimentation? Does extended residence time alter the system chemistry? Have changed upstream conditions increased the possibility of deposits?
Examine the mechanical configuration of the system. Do pipe loops and elevation changes create spots where solids can accumulate? Has selection of minimum pipe sizes led to very low velocities in parts of the system?
Do specific pipe loops have a history of problems? Could these problems worsen at low rate production?
UDC can cause severe damage. Don’t let its occurrence while operating at low rates catch you by surprise. Examine your system to look for UDC possibilities before problems occur.