Fluid Handling: Can Totes Cope?
This Month’s Puzzler
We meter corrosion inhibitor (an aqueous mixture of sodium benzoate and sodium nitrite) into tanker trucks containing our product, ethylene glycol radiator fluid. Previously, we metered the inhibitor into lines to the product tanks. Corporate and our customers can’t seem to decide if they want to continue the current approach or revert to the original system. Complicating this further, corporate is grappling with whether to increase the injection rate by 25%. Now, it’s October in Indiana and winter is coming. “What are we going to do when winter comes?,” an operator complained. Money is tight, I told him.
We use 275-gal totes of inhibitor, and go through 8 or even 12 on some days. Storage space limitations restrict us to only six totes near unloading; the others are stored way across the plant, taking a fork truck about 30 minutes to deliver.
Safety already is upset by the lack of containment around the totes. Because the whole system is “temporary,” we’re using a ½-in. air-diaphragm pump and a ¾-in. garden hose strung 75 ft from the totes outside the dike wall to the loading arm 18 ft above ground level.
Sales is thrilled because the chemical in the totes includes new additives the customer wants. Production is concerned about the additional inventory filling its warehouse and yard — and also displeased with the slow filling rate that adds 20 minutes to the operation and requires an operator on standby because the measurement is by tote level. I’m not happy that we’re wasting ingredients due to all the leaks. I’m not sure how quality control feels about this.
How can we prepare for winter at this late date? Should containment of a water solution worry us?
Assess Multiple Issues
Consider the following:
1. To alleviate storage space problems, think about increasing the concentration of sodium benzoate and sodium nitrate. Doubling the concentration, if feasible (according to solubility), would lower the number of daily tote requirement from 12 to 6. Of course, you also must ensure the metering pump’s stroking requirements or speed allow this, because you only will need half the rate of addition.
2. The 275-gal tote system entails considerable handling, raising the potential for leaks and spills. If space is available, consider installing a tank to eliminate tote handling if this fits in with long-term strategic marketing goals. Obviously, available space and number of deliveries (say, per month) from the corrosion inhibitor vendor will influence tank capacity. Because “money is tight,” this may be a difficult decision requiring careful thought.
3. Because the tote system relies heavily on daily receipts of inhibitor, traffic disruptions or delays could adversely impact business and customer goodwill.
4. The freezing point of an ethylene glycol (EG) solution gradually drops as the glycol content rises. A 60% solution has a -60°F freezing point. Survey your system for water content and dead legs, and consider heat tracing where potential of freezing exists.
5. The problem description gives the impression that most departments don’t favor the tote system. Consider what’s involved in reverting back to the original system. Does it pose any potential production/safety/environmental issues that need addressing?
6. It seems that the switch to the tote system was made in a hurry. For future projects, implement a management-of-change system. This may require diplomacy in “educating” your marketing folks.
GC Shah, consultant
Houston
Implement Realistic Options
You’re in a typical bind for a plant engineer — expected to do miracles with nothing. You have two distinct challenges: 1) preventing freezing; and 2) increasing the pumping rate. The storage and fork truck issue is merely an inconvenience. I won’t get into using automation or a large heated storage tank because you couldn’t get the money for those now.
Try to find some spare warehouse space. If none is available, identify a location where you can hang heavy tarp as an A-frame or expandable modular frame. You probably can’t buy anything ready-made that fits 275-gal totes with enough space. The tarped enclosure should allow for moving totes in and out quickly.
I solved a similar problem at a plant by creating a 2×4 frame with a tarp on a curtain rod so an operator could access an individual tote. I left sufficient room around the totes to allow good ventilation for heated air.
Now, I come to the hard part: heating. You need a portable heat source. Electricity gets expensive to set up and use; so, too, does steam, with the added risk of water creating a slip hazard. I suggest a propane heater providing 24,000 BTU/h for six totes in the enclosure; you’ll use about 3 gal/d of propane per tote at about 0°F outside temperature and 75°F inside temperature. The danger with any kind of heater is fire, so leave plenty of room around the heater and protect it with bollards.
Keep the fittings, hose and pump inside the enclosure. This will protect the pump and shorten the length of pipe needing heat-tracing. (I suggest self-regulated electric tracing.) Moreover, inside fittings will ease the operators’ job of connecting totes.
I derived a crude thawing equation for water to disabuse operations from thinking about that as an option: , the thawing time, h, = 3.07 V0.2061, where V is in gallons (at 75°F room temperature). This doesn’t account for salt freezing-point depression. For a 275-gal tote of water, = 10h. That should convince anyone that thawing totes is a fool’s errand.
Consider temporary blankets and pad heaters for the totes being unloaded directly. Blankets for totes run about $200 each and are hard to unwrap. Bottom pad heaters cost about $1,000 each. Heating with a pad uses about 1,600 W at 115 VAC.
As for increasing the filling rate, first look at the utilities. I assume you’re using an air-operated diaphragm pump because it’s most common for temporary installations. See if the line pressure drops when that pump is running. Watch it for a day; if the pressure is above 80 psig, then you can re-size for a larger pump. The cost is minimal, although you may have to rely on level measurement of the tote instead of flow measurements as the meter may be over-range.
Dirk Willard, consultant
Wooster, Ohio
March’s Puzzler
We use a variety of solvents at our pharmaceutical plant. Until recently, we largely ignored the vent system feeding our thermal oxidizer (TOX). Although the decanting and distillation tower for separating solvents works flawlessly, the TOX system sometimes acts up; the environmental regulators forced us to install a backup system. Lately, the vent system drew our attention when vapor detectors caused a trip during new solvent unloading. Then, everything went haywire. I was sent by corporate to investigate.
The operator claims the suction is very bad and truck drivers complain about the fumes. I found out that, to avoid the fumes, the drivers hook up and walk away and the operator watches from the control station. Plant engineering gripes that a series of expansion projects connected to the system without addressing underlying problems. With budget cuts, corporate says there isn’t any money. One engineer grumbles that the vent alarm is faulty; he says the over-loaded distributed control system suffers from a power supply problem that causes alarm trips.
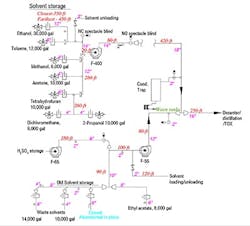
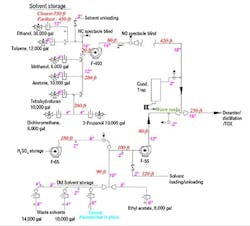
As I expected, the utility piping and instrumentation diagrams are a joke. The line sizes are missing and many vent connections either don’t appear or go to unused tanks. I found a show-stopper: a tank with a blind flange removed from a vent nozzle. Maintenance is pretty sure the slide gate is closed on the abandoned tank and says not to worry about it. I drew up a quick flow diagram to begin my investigation (Figure 1).
Figure 1. Long-ignored system caused intermittent alarm trips and now has gone haywire.
A walkdown of the system showed corrosion and water vapor at some of the flanges, duct joints and two blowers, (F-55 and F-400, the main blower). The other blower (F-65) looks clean because the tank hardly ever is emptied. Maintenance says the corrosion is normal.
Where should I start? What should I do about the abandoned tank?
Send us your comments, suggestions or solutions for this question by February 12, 2021. We’ll include as many of them as possible in the March 2021 issue and all on ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Rd., Suite 400N, Schaumburg, IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you’d like to pose to our readers, send it along and we’ll be pleased to consider it for publication.