Keep Your Cool With Heat Transfer Calculations
[pullquote] Engineers routinely use simulators to evaluate equipment and calculate thermophysical properties. However, you never should take any result as accurate unless it’s tested against measured data, as one recent case exemplifies. It involved investigating the performance of a heat-transfer-fluid system.
The system, which uses a fluid with a proprietary blend of propylene glycol, water and corrosion inhibitors, wasn’t working as desired. One possible cause was deposition of corrosion products.
To understand the problem, first we must consider the real nature of the fouling factor in heat transfer calculations. Conventionally, it serves to make the calculated heat transfer match the observed result. The factor incorporates all the sources of difference — including not just fouling but also disparities between calculated methods and reality, errors in data, and flaws in estimating physical properties.
Most programs calculate single-phase heat transfer in turbulent flow in tubes based on the Sieder-Tate equation:
Nu = 0.023Re0.8Pr0.33(µb/µw)0.14
where the Nusselt number, Nu, incorporates the film coefficient, and both the Reynolds number, Re, and Prandtl number, Pr, contain a number of fluid properties. Properties required include specific heat, density, viscosity and thermal conductivity. Also needed are the viscosity at the bulk temperature, µb, and at the wall temperature, µw; the effect of the viscosity term to the 0.14 power is small, so we’ll ignore it in this discussion.
Simulator packages take two different approaches to these properties. Composition and fundamental relations, often expressed in an equation of state (EOS), can provide some properties. Most models determine specific heat this way. Simulators derive other properties such as density and transport properties, e.g., viscosity and thermal conductivity, from the values for pure compounds adjusted by blending or mixing rules to represent the mixture.
You only should apply an EOS and mixing rules for their intended specific mixtures and ranges. Extrapolation outside the range of applicability can lead to serious errors. To cover a wide variety of conditions, all commercial simulator packages include multiple options for EOS and mixing rules. Unfortunately, many users erroneously assume that method selection isn’t that important. Aggravating this, figuring out the data sources and applicability ranges for the different simulators and their options often is very difficult.
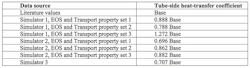
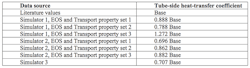
Table 1. The simulator results differed significantly from the one obtained via literature values.
Now, let’s examine the simulator results for the propylene-glycol/water system compared to using the literature values for the fluid properties. Table 1 shows the variation in calculated heat-transfer coefficient.
The base tube-side heat-transfer coefficient came from using literature values provided by the fluid vendor for the properties of the mixture. These were checked by curve-fitting values from the open literature that bracketed the 50/50 composition range. The property values from the vendor and in the open literature matched well.
In comparison, the closest simulator values for system properties result in heat-transfer coefficients more than 10% away from the base value. In some cases, deviations reached about 30%.
Table 2 gives ranges of heat-transfer coefficients for each of the properties. These numbers don’t add up to the ones in Table 1 because some deviations offset each other. Table 2 shows that density errors aren’t that significant but deviations in estimated specific heat are — and that thermal conductivity and viscosity differences are most important.
Table 2. Three of the properties can cause substantial deviations.
The film coefficient is only one part of the heat-transfer evaluation. The total deviation will be less than that shown here but errors of up to 30% can lead to significant misses when evaluating fouling.
What’s interesting is that the simulators mostly predicted lower heat-transfer rates than those using the literature values for the fluid properties. The simulator physical-property estimates lead to a lower predicted fouling than that derived via the vendor-supplied physical properties.
The key lesson here is that, before making a decision based on simulator results, you must understand the methods involved to generate the physical properties, how they differ among methods, how they compare to data, and what that may mean for your conclusions. (For more simulation tips, see “Stifle Simulation Snafus.”)