Polymer membranes with hour-glass-shaped pores combine high permeability with high selectivity to provide significantly better performance than conventional membranes for gas separation, say University of Texas at Austin researchers. Moreover, the thermoplastic membranes remain stable at 450°C and can handle aggressive fluids that can cause problems for other membranes, and their porosity can be tailored for specific applications, notes Ho Bum Park, a post doctoral student at the school. He first prepared the membranes while studying at Hanyang University in Seoul, Korea.
The membranes consist of dense glassy polymers, notes Park. Because these materials lack solubility in common solvents and so arent directly amenable to solution casting, which is the most widely used means of membrane preparation the researchers devised a workaround. They instead form membranes from precursors highly soluble aromatic polyimides containing ortho-positioned functional groups and heat these at 450°C for one hour to cause an irreversible reaction to create so-called thermally rearranged (TR) plastic.
The resultant films are tough and ductile, unlike zeolite and carbon membranes, which are fragile and brittle, the researchers claim. At this point, both self-supporting membranes and coatings on inorganic supports such as alumina and porous stainless steel are being considered; achieving defect-free coating is apt to be easier and simpler than for other materials, add the researchers. While conventional membranes typically have a life of three to five years, 10 years' life is a possibility for membranes made of TR plastic, because of its chemical and thermal resistance, reckons Benny Freeman, a professor of chemical engineering.
The family of TR plastics that can be created, he says, span one to two orders of magnitude in porosity. Varying the precursor and treatment temperature, as well using dopants enable tuning of properties.
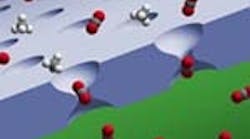
Figure 1. Larger adjoining cavities enhance transport of material to neck of pore, which offers good separation. Source: CSIRO.
The hour-glass-shape of the pores (Figure 1) speeds transport of material into and out of the pores, while the narrow neck provides good separation, notes Freeman. Selectivities for separations such as CO2/CH4 and O2/N2 approach or match those of carbon molecular-sieve membranes. Australia's Commonwealth Scientific and Industrial Research Organization has been involved in analyzing the material and has posted an animation of the separation of carbon dioxide from methane using a TR membrane (see a video presentation here).
The researchers are particularly eyeing the removal of carbon dioxide from natural gas. "If this material was used instead of conventional cellulose acetate membranes, processing plants would require 500 times less space to process natural gas for use because of the membranes' more efficient separation capabilities, and would lose less natural gas in their waste products," says Freeman. "They are excellent for air separation," he adds. Use in fuel cells is another possibility. Freeman also foresees a potential role as a less-energy-intensive way to separate olefins and paraffins.
The first industrial trial, however, is likely to be for removal of carbon dioxide from power-plant flue gas. Park expects to see such an application in Korea within five years.