Save Energy In Distillation
Separations operations account for roughly 50–70% of the energy used in large-scale chemicals manufacturing [1]. Distillation — which we’ll use to encompass the related operations of absorption and stripping — dominates for such separations.
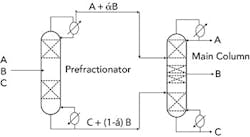
Figure 1. Adding such a column typically leads to a 20–40% cut in energy consumption.
Distillation rules the separation landscape not because of any efficiency advantage but rather because of several other factors that favor it. For instance, distillation:
• scales well, generally to the 0.6 power;
• when performed by itself, doesn’t introduce an extra mass agent (solvent, sorbent, etc.) that needs subsequent recovery, as in the case of absorption or liquid extraction;
• allows heat integration within individual units and across facilities, which can foster effective heat utilization in many separations operations; and
• provides an inter-relationship between pressure and temperature.
Moreover, the technology is well understood and robust, leading to high confidence in designs.
However, its maturity means that distillation usually isn’t the focus of academic research, with several notable exceptions such as the work of Rakesh Agrawal at Purdue, Bruce Eldridge, Frank Seibert and Gary Rochelle at the University of Texas, and Ross Taylor at Clarkson. Fortunately, long-established and robust industrial consortia carry out investigations and continue to refine the practice of distillation. For instance, Fractionation Research, Inc. (FRI), which has more than 85corporate members, operates industrial-scale distillation columns and performs research on modern distillation devices.
Progress is taking place in decreasing distillation energy consumption. Facilities now being planned certainly can benefit. However, energy reductions alone generally can’t justify investment to replace an existing column.
Operating companies expect distillation columns to operate for decades; a life span of 30 years isn’t uncommon. So, sites over time may make modifications and upgrade instrumentation, and often keep the units in excellent physical condition.
Thus, the most-feasible approach for saving energy is via retrofitting. Here, we’ll focus on some ideas for energy reduction in existing columns.
Converting An Existing Sequence
If the infrastructure already exists to perform the separation by conventional methods, you may be able to convert the current sequence to a prefractionator or Petlyuk arrangement (Figure 1). This can save a significant amount of energy for the required separation; typically, it reduces energy consumption by 20–40%. Alternatively, you can produce more products from the facility for the same amount of energy.
Converting an existing facility from a conventional sequence to a prefractionation one certainly isn’t trivial; it requires careful study, including rigorous simulation, and then a detailed evaluation of the mechanical modifications necessary. You should assess, e.g., the internal tower hardware before and after the modifications, suitability of the pre-fractionator reboiler and condenser in the new service, turndown capability in the new service, and other details.
Figure 2. Existing towers with low-volume overheads or bottoms often are good candidates for conversion.
A further way to achieve or increase energy savings is by converting the sequence to sequences that employ side-rectifiers or side-strippers.
Opting For A Single Dividing Wall
A dividing wall column (DWC) may save a substantial amount of energy. (See: “Consider Dividing Wall Columns,”[2].)Depending on whether significant changes in product mix or capacity requirements have occurred over time, it may be possible to convert a two-tower system to a single dividing wall column (Figure 2). This is more likely when the original tower configuration was developed to remove relatively small fractions of light component (A) or heavy component (C) or both.
You must assess mechanical details such as tower hydraulics, feed locations and auxiliary equipment rating as well as other details to determine the feasibility of such a conversion. As with all complex column configurations, it’s essential to remember that the levels of energy consumption are an important consideration for the evaluation of the benefits of new separation configurations. While the single DWC will consume less energy from a First Law perspective, it maybe disadvantaged from a Second Law perspective. So, you must analyze the system individually, taking into account the utilities and economics for the particular operation or entity.
Powerful Low-Cost “Tricks”
Small changes that require minimal capital investment sometimes can produce great benefits. You should consider such “tricks” when searching for improved energy or capacity performance from an existing facility. Some examples include:
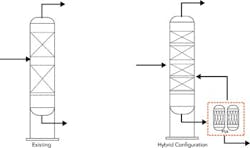
Figure 3. Using a membrane as a prefractionator frequently can provide substantial energy savings.
• Feed location. Always look into this. A feed in less-than-optimum position in the distillation column negatively affects energy use and, thus, capacity. Feed location might have been appropriate initially but, as time has passed and feed or product specifications have changed, may require repositioning. A small change in feed location can save on large reboiler duties.
• Use of side-draws. A precursor of the concepts of prefractionation and the DWC is the thoughtful use of side-draws in a given multicomponent distillation train. A classic example is that where top and bottom products are needed at high purity and the middle boiler is present in small amounts in the feed. If you look into the composition profiles of existing columns such as these, you’ll see that the concentration of the middle boiler presents a steady-state “bulge” that reduces at the ends. Using a side-draw within that bulge allows retrieving the middle boiler at high concentration in a small stream, drastically reducing the heat duty of the column.
• Treating pressure as a variable. Engineers generally think of the operating pressure of a distillation column (i.e., the average between top and bottom) as a given. However, you can effectively use changes in pressure to alter performance. Because relative volatilities vary with pressure, it sometimes proves useful to lower column average operating pressure to require less heat duty. A very common example of this is the well-known trend in refineries to use low-pressure-drop packing in crude and vacuum towers instead of trays to reduce pressure drop and improve fractionation.
• Feed condition. Adding or removing heat from the feed can change the requirements of a reboiler or a condenser. This may not alter the overall heat balance but might allow for easier heat recovery schemes with feed exchangers that may help with the loads on reboilers and condensers.
Properly assessing the viability of these modifications requires a well-validated model of the distillation column that can provide reliable pressure, composition and temperature profiles.
Hybrid Operations
Progress in both membrane and adsorption materials has enhanced the prospects of hybrid separation, by which we mean the use of either membranes or an adsorber in tandem with distillation. The combination of these unit operations potentially can reduce the separation energy of some energy-intensive separations while still meeting the financial hurdles required of any improvement project.
While myriad combinations of other operations with distillation are possible, these two have a strong synergy with distillation that can lead to improved energy efficiency or capacity.
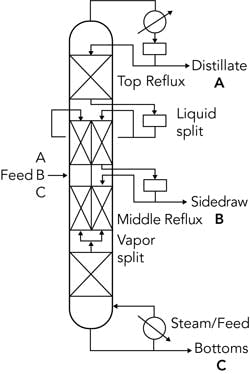
Figure 4. An adsorber can enable reintroducing a light component at an optimum location in a column, lowering energy consumption.
Distillation plus membranes. Membranes are well suited for pre-fractionation; so, they may offer an attractive option for low energy separation upstream of the distillation tower. Figure 3 provides a simplified representation of the concept. (The graphic shows a tower with packing but the concept applies equally well to trays.) The permeate and retentate from the membranes then can go to the optimum location in the tower, lowering the energy consumed by a substantial fraction. As pointed out by Sholl and Lively [3], the use of membranes to pre-fractionate light olefin from paraffin might reduce the energy requirements by a factor of 2 to 3.As always, the key challenge to any large-scale separation with membranes is the surface area required. Membrane separations scale directly, while, as noted earlier, distillation scales to the 0.6 power, which drives large-scale separations to distillation. Applying membranes in tandem with distillation doesn’t change the scale factor but does eliminate the need for the membrane to provide pure product; this enables selecting the membrane area that gives the optimum level of performance within economic constraints.
Distillation plus adsorption. Such a system holds promise for lowering the energy consumption for separating zeotropic mixtures. Indeed, this hybrid operation already is widely used for breaking the azeotrope between ethanol and water — it may well be the preferred method of operating assets producing anhydrous ethanol. As shown in the simplified schematic in Figure 4, the light components are allowed to “slip” to the bottom of the tower, from which they go to the adsorber where light components are captured and returned to the tower at the optimum location.
While the figure illustrates pressure swing adsorption (PSA), there’s no fundamental reason why a hybrid process couldn’t use temperature swing adsorption (TSA).The ability to rapidly regenerate beds with pressure swings, thus reducing the amount and size of the adsorbent beds needed, generally drives selection of PSA over TSA.
Rethink Your Configuration
As we’ve pointed out, retrofitting existing columns with sequences such as Petlyuk processes, DWC, side strippers and side rectifiers and other complex arrangements may conserve distillation energy. In addition, progress in both membranes and adsorption materials has opened up opportunities for applying hybrid unit operations that may well fit into existing infrastructure and produce acceptable economic returns on energy reduction opportunities.
Modern simulation tools and optimization methods enable investigating all the potential alternatives for distillation separation sequences and selecting the best sequences. They play a key role in the design of new facilities but also can help in revamping existing columns where energy savings alone won’t justify a replacement unit. Perform such evaluations for those revamping options that seem appropriate for your plant; you may identify opportunities for substantial reductions in energy consumption.
JOHN PENDERGAST is a professional in residence at Louisiana State University, Baton Rouge, La. DENNIS JEWELL is a fellow at the Dow Chemical Co. in Freeport, Tex. DAVID VICKERY is a manufacturing and engineering technology fellow at the Dow Chemical Co. in Midland, Mich. JOSE BRAVO is president of Fractionation Research, Inc., Stillwater, Okla. Email them at [email protected], [email protected], [email protected] and [email protected].
REFERENCES
1. Adler, S., Beaver, E., Bryan, P., Robinson, S. and Watson, J., “Vision 2020: 2000 Separations Roadmap,” Ctr. for Waste Reduction Technologies, Amer. Inst. of Chem. Eng’s, New York City (2000).
2. Pendergast, J., Vickery, D., Au-Yeung, P. and Anderson, J., “Consider Dividing Wall Columns,” Chemical Processing (Dec. 2008),
3. Scholl, D. and Lively, R.,“Seven chemical separations to change the world,”Nature, 532, p. 435 (April 28, 2016).