A “crumpled” membrane less than 10-nm thick that filters liquids and gases potentially can cut energy consumption in industry, say researchers at Imperial College London. They believe their composite membrane ultimately could improve or replace processes that rely on evaporation and distillation. The membrane, which consists of a polyamide nanofilm on an alumina support, boasts ultrafast solvent permeance and excellent selectivity as well as suitable mechanical strength, they add.
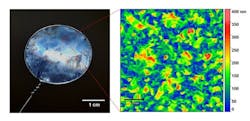
Figure 1. Tiny crumples and ridging at the nanoscale help filter organic liquids under extreme pressures. Source: Santanu Karan, Imperial College London.
“Membranes are currently used for a range of important tasks such as making water drinkable and life-saving kidney filtering. The drawback has been that industry hasn’t been able to use membranes in organic liquid systems more widely because they’ve had cost and design limitations. Our research suggests that we can overcome these challenges, which could make these membranes useful for industries ranging from pharmaceutical companies to oil refining. The energy and environmental benefits could be massive,” notes Andrew Livingston, a professor in the department of chemical engineering at the university who led the project.
Livingston adds that demand for such membranes can be found across all industries where organic liquids are used and contain solutes that must be concentrated or molecules which must be purified.
The crumples on the polyamide nanofilm increase surface area for filtering substances. The membrane remains strong and resists buckling even at extreme pressures; it can filter organic liquids at 50 bar. Moreover, the membrane can withstand a range of organic solvents.
In tests of the 80-mm-dia. prototype membrane, the researchers found that the crumpled membrane only allowed the desired alcohol to pass through, and could separate substances 400 times faster than a conventional membrane. More details appear in a recent issue of Science.
The team believes the membrane can be scaled up to sizes required by industry. “In 2016, we will start [on] A4 [about 8.3 in. × 11.7 in.] sheets, then 30 cm wide and 10 meter long length,” says Livingston.
“We are seeking ways to make the film more hydrophobic so as to have higher flux of hydrophobic solvents, ” notes Livingston. If all goes as hoped, Livingston expects to perform pilot-scale trials of the membrane in 2017.
Other projects involve developing a fabrication process that doesn’t require cadmium hydroxide nanowires, and researching the effect of smooth versus crumpled membranes on fouling; The researchers also plan to run longer-term tests of robustness. Livingston says this work will take anywhere from six months to two years.
However, scaling up the approach to create membranes of a size sufficient for industrial use does pose some issues, he admits, namely, reproducibility of flat sheet membranes, rolling membranes into spiral modules, as well as the cost of components and cost of capital tooling.
Other key challenges to address include fabrication route, scalability and manufacturing feasibility.
“We now want to work even more closely with industry to further refine our membranes so that they can meet their needs. We hope our work will lead to new collaborations and ultimately, improvements in the way industries use separation processes,” adds Santanu Karan, also from the department of chemical engineering and member of the project team.
Livingston says the researchers’ principal partnership is with BP. “We are developing manufactur[ing] further before tying up with membrane makers,” he adds.