An air-fin exchanger is a cross-flow exchanger on the air side. Even with multiple tube passes, getting close approach temperatures is difficult. Air-fin exchangers often pinch out against air inlet temperature on very hot days (see: “Cope with Condenser Constraints”). Against a pinch, higher air flow rates give little benefit — the only effective technique to improve air-fin performance may be to drop the air temperature. Spraying water into the air can do this.
The objective isn’t to put bulk water on the exchanger but instead to create a mist in the air that leads to humidification cooling. So, let’s look at when a mist is useful; how fine a mist is needed; and how to make the mist.
The goal is to create humidification cooling when the water evaporates. Psychrometric charts detail the difference between various relative humidity levels as air becomes more saturated. The charts include dry bulb and wet bulb temperatures. The dry bulb temperature is the starting air temperature. The wet bulb temperature is the achievable cooling by saturating the air. A quick glance at a psychrometric chart shows very little temperature drop is possible once air gets to ~85% relative humidity. To cool air at 115°F to 110°F requires starting with 70% humidity and increasing the humidity to 83%. Considering a 5° cooling as the minimum performance to make the expense worth the effort, a site should have a relative humidity of 70% or less. Checking the required temperature drop against site conditions will clarify if mist cooling might make sense.
[pullquote]
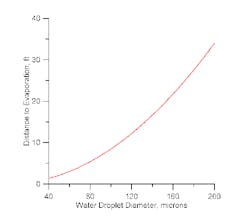
The benefit of humidification comes from evaporating water upstream of the air-fin — by creating a mist immediately underneath the air-fin. Only a short distance is available for the water droplets to evaporate before they enter the exchanger. Based on observation of operating misting systems, my own rule-of-thumb is to get a spray pattern that creates a nominal 50-µ (or smaller) droplet at 3 ft below the air-fin bundle.
ANDREW SLOLEY is a Contributing Editor to Chemical Processing. You can e-mail him at [email protected]