Rethink High-Temperature Materials Processing
Optimizing energy efficiency during high-temperature materials processing seems a worthwhile goal to strive for in developing a new production line. But what does it mean? And where do you start? To consider the scope of opportunity for energy efficiency to reduce cost and improve operations, it's important to pinpoint a few baseline principles.
First, the initial development of an advanced material process usually focuses on the technical value of the product, not the elegance or efficiency of the process. The material's end physical properties drive development. Energy efficiency gets considered later, when product cost begins to become a significant concern. In a worst-case scenario, the production route developed in the laboratory can hinder true efficiency as the process is scaled up to industrial levels.
Second, high-temperature processes are notorious wasters of energy — as exemplified by the hot working environment around steel forges and aluminum smelting pots. Those processes are modest compared to ones for some cutting-edge materials that operate at 2,000–3,000°C. The opportunity for improvements in efficiency is great but these come at a cost or a sacrifice.
THIMBLES TO TONS
The road from process inception to commercial production is arduous. Scaling of thermal processes rarely is a simple matter of linear extrapolation.
At experimental scale, the conversion rate of many solid/solid and solid/gas reactions primarily depends upon the setpoint temperature, overall atmospheric chemistry, size of the reactants and the quality of the intermixing. In small research test furnaces, the furnace temperature can track the control profile very well. The sample load, if relatively small, also may track the desired profile well. At the same time, the small internal volume of the furnace and ratio of sample to furnace volume simplify removing product gases and replenishing with fresh gas. Under these experimental conditions, product uniformity is rarely a significant concern.
As the reaction is scaled to larger sizes, the ability to heat or cool the mass of material and introduce or remove gases from the solids plays an increasingly important role in reaction efficiency. Often these factors become the primary variables that control the conversion rate. They can create limitations that extend the total processing time, impacting both throughput and total energy utilization.
Moreover, batch processing, if chosen for production, takes a further, significant toll on energy efficiency. The requirement to heat and then cool the entire product load, the reaction containers, structural components and all the refractories is especially inefficient. In cases demanding higher temperatures and faster heating/cooling rates, water cooling may improve the functionality of the equipment — but at a substantial sacrifice in energy efficiency.
MOVING BEYOND BATCH
The most efficient thermal processes are engineered to apply heating and cooling to the smallest load possible. In a perfect situation, this means processing the reactants in a continuous manner without need for material containment. The three most critical design criteria that affect efficiency are reactor type, refractory and atmosphere management. So, let's look at each of these.
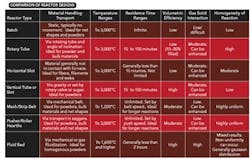
Reactor type. During scaleup, it's crucial to select the most appropriate reactor. Table 1 lists some characteristics of various types of reactors. Each design brings process advantages and challenges. Often a material's handling characteristics and behavior during processing will dictate choice of a certain type of reactor. Additionally, the need to mix material while it's heated can preclude the use of a reactor where the material is constrained or stagnant.
Conveyance furnaces come in a wide variety of designs and materials of construction. The highest efficiency units are ones that recover heat or recirculate hot containers within the heating zone of the furnace, thereby limiting heating and cooling requirements almost exclusively to the process material. In more-advanced designs, heat recovery from product carriers can boost system efficiencies further. Sometimes, it's even possible to engineer material flows so cool incoming reactants get preheated by exiting material. However, these designs are less common and can pose design and operational difficulties. Nonetheless, they can provide the best efficiency for processes demanding container-supported heating.
Gaining much more energy efficiency requires eliminating the need for containered material conveyance altogether. One option is a rotary tube furnace. As the reactant bed moves through the tube, it's constantly stirred by the tube rotation. This stirring action boosts thermal transfer to the bed, improves removal of product gases, and increases solid/gas exchange in cases where the furnace gas also is a reactant. These enhancements often lead to product with better uniformity than materials processed in a static bed. Additionally, because only the reactant powder is heated and cooled, thermal efficiency is significantly better than that of pusher-style furnaces.
Vertical furnaces offer a reasonable alternative to rotary tube furnaces in cases where material movement in the rotary furnace is unsatisfactory or where other features such as very short or very long lead times, significant interaction with reaction atmospheres or completely contact-free reactions are required. Similar to rotary furnaces, energy use primarily is related to heating of the product and supporting necessary reactions and, thus, is relatively efficient. The design allows for minimal interaction with the furnace wall, making it a good choice where contamination is an issue. In some cases, this type of furnace is invaluable in combination processes, e.g., spray pyrolysis coupled with calcining.
Refractory. The selection and design of refractory can significantly impact thermal efficiency. In batch systems, highly efficient refractory designs may save energy and boost ramp speed during heatup and soaks but lead to extended cooling times and, thus, longer total cycle times. Less efficient designs may improve the cooling rate but pose shell-temperature or other limitations. Alternatively, water-cooled equipment may provide a reasonable solution for cooling rate and minimize space and weight requirements for refractories — but will dramatically decrease energy efficiency.
Continuous processes decouple the heating, soak and cooling portions of the process by distance. Therefore, using different refractory designs along the length of the furnace can optimize the thermal management and thermal efficiency requirements in each stage.
Atmosphere management. Heat losses via exhaust designs also significantly differ between batch and continuous equipment. The ability to utilize waste heat generated by batch furnace exhausts is limited. Because the waste heat in such equipment essentially is at the temperature of the load, there's no opportunity to insert the heat value directly into the process.
In contrast, continuous processes routinely provide a variety of options for heat recovery. For instance, counterflow gas can help cool product leaving the process and subsequently help preheat the incoming reactants. Also, gas composition at a given position in the furnace is more predictable. In cases where incoming gas chemistry or removal of gaseous products influence the reaction rate, this predictability enables minimizing gas usage and, therefore, thermal losses related to the gas.
Where the exhaust gas contains waste products having fuel value, advanced equipment design can combust the gas and use the hot stream for preheating the primary process. In some cases, such waste-gas streams can provide very substantial fractions of the total thermal requirement for a process. The result is significant energy efficiency and reduced associated costs, as well as lower costs and environmental concerns compared to alternatives for abating the waste-gas flow.
INSIGHTS FROM CARBON FIBER
Conversion of polymeric fiber materials to carbon fiber typifies how processes evolve. Large-scale production of carbon fiber began in the 1970s. Initial equipment was less than 1,000-mm wide and produced under 100 tons per year — and required an input of 150–200 kWh per kilogram of carbon fiber.
Through successive scaleup efforts, the state-of-the-art for conversion of carbon fiber now has advanced to a system width of 3,000–4,200 mm and single-facility production rates exceeding 2,500 tons per year. Through increased reuse of energy (integration of waste energy to drive the process), equipment improvements (via economies of scale and refractory advancements) and better basic feed material (polymers with higher conversion to carbon fiber), the specific energy consumption is now less than 20 kWh per kilogram of carbon fiber. Figure 1 highlights the progressive reduction of energy requirements through the scaleup process.
In many ways carbon fiber production can serve as a useful guide for improving the efficiencies of other thermal processes. Polyacrylonitirle-based carbon fiber production is performed in an entirely container-less mode; the fiber itself provides the motive force to move the product through subsequent process steps. This provides the energy efficiency of a continuous process, augmented by further gains from eliminating the need to heat ancillary support equipment. The only system inefficiency is the heat loss related to the cover gas used to maintain the appropriate atmosphere. Development efforts to reduce this atmosphere loss through improved furnace end seals are ongoing.
POWDER PROCESSING OPPORTUNITIES
Most thermal processes involve bulk materials in the form of powders or aggregated mixtures that require some type of container or construct, such as a belt conveyor or rotary tube, to assist in material transport through the process. Minimizing the energy losses associated with this aspect of the equipment design is one of the most significant opportunities for improving energy efficiency. Design optimization often involves concern for this energy efficiency in concert with considerations for enhancing reaction kinetics, gas exchange, residence time and materials of construction. Therefore, the requirements related to a specific process will drive the options for production methods and, thus, the efficiencies of that process.
In practice, rotary tube furnaces provide notable values of throughput and efficiency for a wide range of powders, as long as the materials behave properly during the process. In some cases, the nature of the reactant material can cause flow problems. Powder adhesion to the tube can change bed mixing behavior, flow through the furnace and, in extreme cases, cause wide swings in material residence time. In other cases, material entrainment in the exhaust gas can affect throughput and flow. Sometimes changing the reactants' physical form — e.g., granulating fine powders into pellets or aggregates — can overcome these problems. Alternatively, the use of internal features within the process tube can promote desired bed behavior. When the nature of the material doesn't allow for granulation or the granules aren't sufficiently strong to retain their shape throughout the process, other means of high throughput and high efficiency processing are needed.
EMERGING OPPORTUNITIES
Recycling the process heat from the product during cooling promises to further enhance energy efficiency. Transferring that heat to incoming reactants means less energy must be inserted into the process. It also can foster more-compact designs because the equipment's heating and cooling portions may be shorter and the heating elements and support equipment may be smaller. Introducing this form of energy efficiency into bulk materials processing equipment is still in its early stages. However, equipment designs that provide this type of heat transfer for select processes have been demonstrated and additional opportunities to expand the concept to a wider range of materials are being investigated.
Empirical testing at a demonstration semi-continuous level provides a proven means to extract scaleup parameters. Scaleup data from a well-designed set of experiments on semi-continuous or continuous furnaces can serve to redefine the process recipe and identify the continuous commercial-scale device that provides the best balance between product quality, throughput, energy efficiency and operating expenses.
TOM MROZ is director of technology for Harper International, Lancaster, N.Y. ROBERT BLACKMON is vice president of integrated systems for Harper International. E-mail them at [email protected] and [email protected].