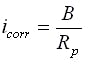Innovative corrosion monitoring solutions enhance process optimization
Introduction:
The purpose of corrosion monitoring has changed dramatically from the early days of weight loss coupon exposure. As new technologies have evolved, so the accuracy of data and relevance of the information to process control has improved.
Corrosion measurement methods in use today still include weight loss analysis, although this is regarded as more of a retrospective status check than a means of 'monitoring'. Off-line measurement methods that interact with the process environment include such general corrosion measurement techniques as electrical resistance and linear polarization resistance. These systems are able to operate in stand-alone mode providing 'spot' corrosion data via battery-powered, field-mounted instruments. The flexibility of installing these systems in remote locations is somewhat offset by the fact that data is available only periodically and there is an overhead in personnel time to download data. Post processing of the corrosion data is often performed in computerized spreadsheet or workbook format.
Contrary to popular belief, though, corrosion occurs neither continuously nor at a uniform rate. The occurrence of corrosion can have a significant adverse effect upon the availability and reliability of plant. Of greater benefit to the plant engineer or process control specialist, therefore, is general and localized corrosion data that can be made available on-line, and in real-time. The ability to interface corrosion data with a plant or process control system effectively takes corrosion to the higher level of being 'another process variable'. This, then, affords the opportunity to both look at corrosion data in real-time and to correlate corrosion activity with changes in other process variables. Enabling the process operator to view all data through the same process control system interface means that he/she is now able to see immediately the effect that process changes have on corrosion activity. The control room engineer also, therefore, has the ability in the longer term to achieve optimum production rates while protecting plant integrity (e.g. minimizing downtime for repair of damage and increasing equipment service life).
There are many positive aspects to the scenario of on-line, real-time corrosion monitoring for process control. In the following discussion we review the methods of corrosion measurement in use today, and discuss their relative merits in affording the user a better understanding of the relationship between corrosion and process variables.
Corrosion Measurement and Monitoring:
The term “Corrosion Monitoring” is often used to describe a broad range of techniques used to evaluate the degradation of metallic materials. These techniques can be divided into two distinct groups, namely those providing indications of the cumulative damage sustained (off-line, retrospective) and those providing indications of the prevailing corrosion rate (usually on-line and continuous). The techniques described below are applicable with virtually all metallic materials including, for example, carbon steels, stainless steels, duplex and super duplex steels, copper and nickel alloys and the more noble metals commonly found in chemical process equipment such as titanium and zirconium. Generally, if a metallic material can corrode then its corrosion behavior can be measured or monitored using an appropriate technique. However, the following practical points must be considered in relation to the type of measurement or monitoring technique that will be used:
a) Fabrication of the required sensor configuration from the selected material - can the required phase of the metal be formed into an electrode ? will the electrode manufacturing process alter the heat treatment of the metal ?
b) For a given metallic 'mixture' (e.g. solder) or de-alloyed material , the exact composition may be indeterminate and so may present slight errors in calculations of corrosion rates.
A good and experienced probe supplier is an important ally in the selection of probe design.
Although all techniques can be applied successfully for almost all metallic materials, the selection of the correct technique is really determined by the type of corrosion to be monitored. When choosing a corrosion measurement or monitoring technique, it is important to consider the why, where and how - some of these issues are considered more fully toward the end of this article and are summarized here:
1. Why do I need to know about corrosion in my process ? Hopefully, your response is that you need "to work out what is causing the corrosion and reduce or prevent it". However, a deeper consideration of corrosion and its consequences will generally help you to work out your measurement needs such as the frequency of measurement required, the level of corrosion detail needed. Some thoughts may include:
a) Corrosion was the cause of an unplanned shutdown - I need to understand what in the process caused the corrosion.
b) We replaced some of our piping last turnaround but it has corroded through again - we need to know what to replace it with next time.
c) We found some pitting corrosion last inspection but we don't know if it was caused by a process upset or something else.
d) Our equipment is halfway through its planned design life but we still have 75% of our corrosion allowance left - we need to monitor to be assured we can extend the equipment lifetime.
e) We are designing a new process and we need to be sure we have selected the correct materials.
2. Where do I need to measure the corrosion?
a) For existing plant with a history of inspection and maintenance data, the location of the corrosion probe can usually be worked out easily. However, if an existing probe access port is not available you may need to spend time compiling a technical case to justify introduction of the new measurement equipment.
b) For new plant, it is wise to consult with a corrosion or materials engineer who can advise on optimal probe locations. Whilst comprehensive monitoring is a useful thing to have, it is important to consider cost-efficiency and ensure that the corrosion measurement in itself will deliver value rather than represent a cost sink.
3. How do I measure the corrosion ? Hopefully, going through the above exercises will have drawn you to a short-list of candidate techniques and sensors. The following descriptions are necessarily brief, but provide an introduction to the types of monitoring methods that are available and hopefully will assist in an initial review of appropriate techniques for your own corrosion needs.
• “Cumulative Loss" techniques, including: Weight Loss Coupon, Electrical Resistance (ER), Thin Layer Activation (TLA), Field Signature Method (FSM), Ultrasonic Thickness (UT) measurement and other non-destructive examination methods (e.g. radiography).
• “Corrosion Rate" techniques, including: Linear Polarization Resistance (LPR), Harmonic Distortion Analysis (HDA), Electrochemical Noise (ECN), Zero Resistance Ammetry (ZRA), Electrochemical Impedance Spectroscopy (EIS), Inter Modulation Distortion (IMD), Potentiodynamic Polarization
Cumulative Loss: The Cumulative Loss techniques will only show some signs of change when sufficient corrosion has been sustained to cause a change in the 'bulk' material properties. As such, most are used off-line and do not provide real-time data. The most popularly employed of these measurements for field corrosion evaluation are as follows:
For conventional ER the sensitivity of measurement is typically 0.1% of the element thickness (similar limitations apply to TLA, FSM and UT techniques). Such methods are most appropriate to use where the corrosion rate is relatively stationary, i.e. the rate does not suffer from large excursions. As accounting tools they are useful in providing an indication of the cumulative wastage of a resource, and hence the likely useful remaining lifetime. Overall, the techniques are relatively insensitive to localized corrosion.
Corrosion Rate: The Corrosion Rate techniques have a much higher resolution and have been developed to provide a fast appreciation of the electrochemical rate processes taking place at the metal/environment interface. Measurements using these techniques may take only a few minutes.
The corrosion current is a consequence of the corrosion process and its value is directly related to the rate of the metal loss. The electrochemical monitoring methods have been developed specifically to estimate the corrosion current. If the corrosion process is relatively stationary i.e. at a “steady-state” then the relationship between the polarization resistance and the corrosion current is given by the Stern-Geary relationship;
where Rp is the polarization resistance, B is the Stern_Geary constant and Icorr is the corrosion current (which is directly related to the rate of metal loss or corrosion).
(Note that the Stern-Geary constant is a system, not a universal constant)
This approximation is the basis for the use of LPR and EIS for the estimation of general corrosion rates. In the case of the LPR measurement the polarization resistance is a simple function of the applied voltage and the current reponse at low frequency. By comparison, the EIS technique is used to determine the characteristic impedance of the corroding metal, typically over the frequency range 1 millihertz to 100 kilohertz.
Harmonic Distortion Analysis (HDA) and Inter-Modulation Distortion (IMD) techniques also rely on a steady state approximation, but require more mathematical treatment than the simple LPR.
For the HDA the current response to a low frequency voltage sine wave is analyzed in terms of the fundamental response and the higher harmonics, to provide values for the corrosion current, the characteristic anodic and cathodic coefficients, and hence a value for the Stern-Geary constant.
The IMD technique is slightly different in that a composite signal comprised of two superimposed sine waves is used to polarize the system. The current response is analysed for the inter-modulation distortion products of the of the two sine waves. This leads directly to the estimation of the corrosion current and the characteristic anodic and cathodic coefficients, and again a value for the Stern-Geary constant.
LPR, EIS, HDA and IMD are powerful techniques developed originally for short-term laboratory evaluation of metallic corrosion, and are finding increased use in field applications for the determination of general corrosion rate behavior.
With knowledge of icorr, the corrosion rate (CR) is calculated according to:
CR = icorr x seconds per year x atomic mass x electrode area
No. of electrons transferred x Faraday's constant x density
The corrosion rate is expressed in either mils per year (mpy) or millimeters per year (mmpy), according to preferred units of measurement.
Electrochemical Noise (ECN) was initially developed to fill a huge gap left by the more conventional techniques discussed above, particularly the area of identifying when localized corrosion phenomena (such as pitting, crevice corrosion, stress corrosion cracking etc) are occurring. The “steady state” analogy for these phenomena is not applicable, since the localized attack occurs in a stochastic, or random, manner. Electrochemical noise is used pro-actively to identify periods when the corrosion processes become unstable, and to recognize when the probability of localized corrosion is high. ECN differs from LPR in that no electrical perturbation of the electrodes is required. The technique measures small, naturally occurring variations in current (current noise, typically microamps) and potential (potential noise, typically millivolts). When general corrosion is occurring the potential and current fluctuations can be processed to provide a general corrosion rate using a similar data treatment to that outlined above for LPR except using a resistance noise (RECN) in place of Rp. When localized corrosion phenomena occur, the characteristics of the fluctuations change dramatically, and these characteristics can be used to identify the most probable mode of attack. From a plant corrosion control perspective, this is important, since the technique provides an early warning of incipient localized corrosion.
Electrochemical Noise has been widely applied to the evaluation of localized corrosion phenomena (pitting, crevice corrosion, environmentally assisted cracking, microbiologically influenced corrosion, etc.). Localized corrosion can occur on many metals, in fact surveys have shown that some 70-90% of all corrosion failures are due to localized corrosion. The difference between general and localized corrosion is in the distribution of anodic and cathodic sites on the metal surface. In the case of general corrosion, the distribution is quite even. Localized corrosion results from situations where a local anodic area can be supported by a relatively large cathodic area. Examples may include where a local breakdown in a protective film occurs on the metal surface, e.g. in passive materials, where scale has partially formed, where a local coating holiday occurs. The potentially catastrophic nature of localized corrosion is realized often through the appearance of pinholes in the metal which, e.g. in the case of pressurized systems, can lead to explosive failure of the equipment.
It is important to note that the electrochemical evaluation of localized corrosion is necessarily limited to a qualitative assessment of the corrosion mechanism, as opposed to the quantitative rate information available for general corrosion attack. This is because it is impossible to predict the number, size and distribution of pits or cracks. Accordingly, a pitting probability or 'Pitting Factor' provides an easily understood representation of localization, where a low value close to 0 (zero) represents general corrosion and a high value closer to 1 (one) represents that localized corrosion is occurring.
Electrochemical noise data has been studied for more than twenty years across a broad range of industrial field applications and a comprehensive assessment of the localized corrosion data resulted in a preferred method of statistical evaluation, namely skewness and kurtosis. In brief, these techniques allow the distribution of current noise and potential noise data to be analyzed statistically as compared with the normal distribution of signals anticipated in the case of general corrosion. The kurtosis shows whether the distribution is more peaky or flat than 'normal', whereas the skewness indicates a shift of the distribution to the left or right of 'normal'. Further information about this data appraisal can be obtained from Reference 4.
Taking Electrochemical Monitoring Into the Field: Some of the considerations to be given to migrating electrochemical techniques to a plant environment are:
1. The choice of a suitable location to insert a probe. Ideally the corrosion measurement should be made in an area where the corrosion processes will be most active, such that the “worst case” scenario is covered. Occasionally this may present some difficulties, particularly in case where the corrosion zone may fluctuate as a consequence of local conditions, for example in condensing environments.
2. The type of probe to use. What will be the maximum operating temperature and pressure? Are the insulating materials suitable for the environment? Can a conventional “off the shelf” finger style probe be used, is a flush mounted probe or a “flow through” probe more appropriate? What exposed surface area of the material should be used (this has a direct impact on the sensitivity of the measurement techniques – low corrosion rates require larger surface areas exposed than high corrosion rates). Are there special temperature control requirements? Consideration should be given to the expected probe lifetime, and the complexity of the design of the probe (the more robust solutions are usually the simplest).
3. Identifying the most appropriate automated monitoring technique Is there a requirement for quick response? Is localized corrosion an issue? Can the measurement be made autonomously on a regular basis without operator control? Fortunately the electrochemical techniques are generally suited to providing a measurement within a period of minutes, the issue really becomes one of quality of data.
LPR is, at first sight, probably the easiest to implement but requires some methodology for minimizing the effects of noise in the corrosion process. It does require some default value of the Stern-Geary constant to be applied. Field proven. HDA has the advantages of allowing derivation of the Stern-Geary factor and corrosion rate directly, but at low corrosion rates, and in unstable systems, the value of the third harmonic (which is required for correct analysis) may be too low to measure accurately or may be corrupted. Field proven.
IMD requires the analysis of the distortion products which are products of the second harmonic (which will not exist if the anodic and cathodic coefficients are equal) and has not been field proven.
EIS difficult to automate interpretation and subsequent corrosion rate evaluation. Not field proven.
ECN is the only choice if detection of localized corrosion is an issue. Field proven.
Monitoring of some forms of localized corrosion (e.g. stress corrosion cracking) benefits from a probe configuration that reflects the physical attributes of the equipment being monitored (i.e. including applied stress or artificial crevice on the working electrode). Also, it is important to note that some forms of corrosion (e.g. erosion or cavitation) are 'physical' phenomena although the effects of them can be detected electrochemically.
4. Assuring data integrity Need to ensure that the data is valid by integrating automated system checks and calibration where necessary.
5. The response (or cycle) time of the measurement The measurement and analysis time frame determines the data update rate to the DCS. Ideally this should be on a similar time frame to other inputs such as temperature, pressure, flow etc, in order that the corrosion information may be correlated with process operations.
6. The ability to provide general corrosion rate information The instrumentation should provide high quality general corrosion information using techniques which will reject adventitious noise such as may arise from mains voltage power lines.
7. The ability to provide information regarding localized corrosion propensity localized corrosion is more insidious than general corrosion, since only a small area of the metal is undergoing metal loss. The localized corrosion rate may be orders of magnitude greater than that being reported by general corrosion measurement techniques, hence it is important to be aware of the transitions between general to localized corrosion.
8. Measurement and analyses automation One of the major difficulties with migrating the corrosion measurement techniques from the laboratory to the field is in automating the measurement and analysis routines. Currently this may be achieved using embedded processors and sophisticated algorithms residing in firmware which control timing and communications, along with measurement and analysis routines, with a high degree of accuracy and precision. The algorithms required for the electrochemical measurements are somewhat different to those required, for example, when measuring temperature or pressure, where the physics of the sensors used is quite well understood and defined. With the electrochemical methods we are trying to determine both the kinetics and mechanistics of the corrosion processes occurring at the metal surface.
9. Simplicity of installation Installing the instrument should require minimum effort in terms of its location, wiring and set-up.
10. Interfacing with the DCS system Ideally the corrosion information should be made directly available to the operator via the DCS to be viewed as another process variable. When integrated into the DCS and the plant historian database, the corrosion data can be analysed with respect to its interaction with other process variables.
11. Convincing the site personnel that the corrosion measurements are a true reflection of what is happening in the system Plant inspections are a relatively infrequent occurrence, usually coinciding with routine shutdown and maintenance. In the real world corrosion rarely takes place at a steady rate for prolonged periods, but usually occurs with some short periods of very aggressive attack and relatively long periods of little or no attack. For example, if the corrosion processes occur for 10% of the time, the actual corrosion rate instead of being say 10 mpy may peak at ~100mpy or much higher. These periods of high corrosion rate often go unnoticed until the events leading to the corrosion increase in frequency and ultimately lead to failure. Subsequent failure analysis finds “corrosion” to blame but does not give any clues as to what happened and when.
System Configuration: The configuration of the system can take many forms, depending upon the intended application. For laboratory studies, a relatively simple layout can be used with bench experiments and flow loops. In the case of field studies, a variety of configurations is available to suit plant installation (hardwire connection to computer or direct to control system) or remote applications where radio communications and/or solar power sources are required.
Probe Configuration: The corrosion probe design is all-important to the system as this is the component that interfaces directly with the process environment and must be both suitable for the installation location and enable representative corrosion measurements to be made. All too often, the quality and relevance of the corrosion data measured can be severely compromised by inappropriate probe design or even installation of the probe in a location where it is impossible to capture the process conditions that actually cause the corrosion.
Integrating with Plant Management Systems for Unified Corrosion and Process Control
The value of data can only be truly realized at the point it becomes information that can be acted upon directly. The benefits to the plant of achieving this include increased unit runtime, also realizing cost savings from decreased inspection and maintenance. By correlating corrosion data with relevant process variables (e.g. temperature, chemical feed rates) and according to the same time base, it becomes possible to establish the root cause(s) of the corrosion. This affords the Plant Operator the possibility of mitigating or avoiding the corrosion by implementing the appropriate process controls. Ultimately, then, corrosion information requires to enter the process control domain in order to become a component of unified plant asset management. The key to a successful systems integration is in providing data at the right level of complexity and according to the desired communications protocols - this enables a direct transfer of data to control systems and the possibility of rapid decision-making by either automated or human response.
Conclusions:
• Corrosion measurement and monitoring techniques exist that have field experience for more than twenty years, and include off-line retrospective methods as well as on-line, real-time techniques
• A key objective for any corrosion management or mitigation program is to detect and resolve corrosion problems before significant equipment damage occurs
• The more advanced corrosion management programs are able to utilize on-line, real-time electrochemical corrosion monitoring to help correlate corrosion with processes and operations
• These newer technologies (e.g. SmartCET®) are proven in monitoring corrosion behavior in real time, even in the harsh world of chemical and petrochemical plants and hydrocarbon pipelines
• The technology works equally well in primarily organic process streams with low water content as with predominantly aqueous streams, although probe design is key to successful monitoring
• By integrating on-line, real-time with process information management systems, Operators and Engineers can view corrosion data in real-time along with the other important process parameters like flow rates, temperatures, pressures, and liquid levels
• The real value proposition lies in achieving sustained process performance optimization by keeping corrosion to a minimum while maximizing equipment uptime, production rates, product yield and plant safety
References:
1. "Real-time Corrosion Monitoring for Improved Process Control: A Real and Timely Alternative to Upgrading of Materials of Construction", Eden, D. C. Eden & Kintz, J. D., Paper #04238, NACE Corrosion 2004, New Orleans, March 28-April 1, 2004.
2. "Real-time, On-line and On-board: The Use of Computers, Enabling Corrosion Monitoring to Optimize Process Control", Eden, D. A. Eden & Srinivasan, S. Paper #04059, NACE Corrosion 2004, New Orleans, March 28-April 1, 2004.
3. "Electrochemical Noise: The First Two Octaves", Eden, D.A., Paper #98386, NACE Corrosion 1998, San Diego
4. U.S. Patent No: 6,264,824 "Assessment of Corrosion", Reid, S.A. and Eden, D.A.
5. "Making Credible Corrosion Measurements - Real Corrosion, Real Time", Eden, D.C., Cayard, M.S., Kintz, J.D., Schrecengost, R.A., Breen, B.P. and Kramer, E., NACE Corrosion 2003, San Diego, April 7-11 2003.