Wood-based Chemicals Get Boost
An ionic liquid (IL) may hold the key to converting wood into biofeedstocks and biofuels in an eco-friendly way, believe researchers in Northern Ireland and Alabama. They are jointly developing technology that overcomes environmental issues posed by the 120-year-old kraft process. (For more on developments in biofeedstocks and fuels, see "Biofeedstocks Still Grow," www.ChemicalProcessing.com/articles/2009/094.html.)
BASF, Ludwigshafen, Germany, is sponsoring the work being conducted by Dr. Héctor Rodríguez and Professor Robin Rogers of the School of Chemistry and Chemical Engineering, Queen's University Belfast, Belfast, Northern Ireland, and scientists at the Center for Green Manufacturing and Department of Chemistry at the University of Alabama, Tuscaloosa, Ala. Their efforts focus on the IL 1-ethyl-3-methylimidazolium acetate ([C2mim]OAc) and wood chips from the softwood southern yellow pine and the hardwood red oak. BASF already has licensed some patents arising from the project.
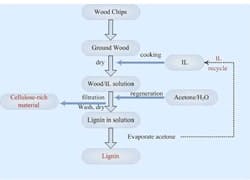
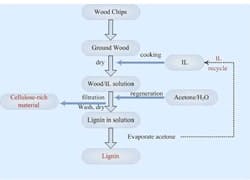
The process itself involves milder grinding of wood chips than in the kraft process, followed by overnight drying at 90°C. The wood mass then is mixed with [C2mim]OAc and heated in an oil bath at 110°C for 16 hours. Centrifugation separates liquid containing dissolved wood from residue. Adding a water/acetone mixture to the liquid releases cellulose-rich materials and lignin in separate phases, without the severe conditions and toxic chemicals needed in the kraft process. Further details appear in the Journal Green Chemistry.
"We think that this technology can revolutionize the way in which biorefineries are understood nowadays. The ionic liquid technology should allow for separately yielding the major biopolymers and possibly even other minor components of high value in wood and other biomass. Further processing, possibly with ILs, could yield a plethora of materials and chemicals, either as final products or as feedstock for other industries. This would be achieved through an environmentally friendly process, with reductions in the generation of waste or the consumption of energy — opening a door to the eventual development of a truly sustainable chemical economy," says Rodríguez.
In the meantime, the University of Alabama team is continuing experimental research on the use of additives as catalysts for rupturing key chemical bonds to get better yields from the process in a single step.
"The particular challenge consists, in our opinion, in a more efficient breakage of the chemical bonds which link the different biopolymers of wood in its complex structure," explains Rodriquez. "The IL on its own works quite well in disrupting other chemical interactions and dissolving the different biopolymers but additives may assist in better breaking the bonds between polymers, which presumably will end up in faster dissolution with better — perhaps total — separation."
The relatively easy steps and mild processing conditions involved should facilitate development of pilot plants, Rodriguez believes. However, he also feels more investigation of the use of additives is required before commercialization issues can be tackled.
Lack of toxicological profiles can impede commercial application of ILs. Interestingly, this doesn't apply to [C2mim]OAc because BASF already has been using the chemical in its own wood dissolution projects and has characterized its toxicological properties.