Biomass Breakthroughs Beckon
Some processes using biomass-based raw materials already operate at commercial scale, others are near (see "Circular Chemistry Spins Faster,") while new research gives a hint of what the future might hold.
For example, combining genetically engineered bacteria, kraft pulp and an innovative macromolecular design approach to organic chemistry, researchers at the Japan Advanced Institute of Science and Technology (JAIST), Ishikawa, Japan, and the University of Tokyo have developed what they say is the highest heat-resistant plastic ever based on biomass.
Stable to over 740°C, the novel plastic uses no heavy inorganic fillers and is lightweight in nature.
[pullquote]
Producing bioplastics from renewable biological resources is a prerequisite for achieving a circular and sustainable society, but current bioplastics are mostly heat-sensitive aliphatic polymers with limited uses. At the same time, developing aromatic heterocyclic monomers from biomass is problematic because controlling their structure is difficult.
So, the team first genetically engineered bacteria to produce from the renewable kraft cellulosic feedstock two aromatic aminobenzoic acid monomers: 3-amino-4-hydroxybenzoic acid (AHBA) and 4-aminobenzoic acid (ABA).
AHBA was then chemically converted to 3,4‐diaminobenzoic acid (DABA). Subsequently, poly(2,5‐benzimidazole) (ABPBI) was obtained by the polycondensation of DABA and processed into a thermoresistant polybenzimidazole (PBI) film.
Figure 1. The development strategy for ultra-high thermoresistant and flame retardant cellulose-derived PBI and PBI/PA films. Source: JAIST.
The researchers found that copolymerizing DABA with a small amount of ABA increased degradation temperatures of the PBI film to over 740°C.
Results showed ABA strengthened the interchain hydrogen‐bonding of the PBI bioplastic; this was key to its thermoresistance, which the researchers say is higher than any existing biomass‐ or petroleum‐derived plastics, including polybenzazoles.
Further, they believe the bioplastic’s processability enables it to be hybridized with metals and inorganic compounds at temperatures above their melting points without thermally degrading the contacting plastics.
This, in turn, could enable manufacturing engine components that improve energy efficiency for hybrid electric vehicles sensitive to weight. Moreover, PBI bioplastics could be inserted as high‐thermoresistant insulators between conducting metals to create high‐performance electronic devices, including super‐integrated circuits, mass memories, and high‐power motors, opening the door to a new era of lightweight materials.
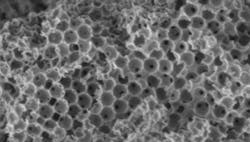
Figure 2. Sponge-like catalysts could transform biodiesel production and chemical manufacturing. This image shows the porous ceramic sponge fabricated in the study, magnified 20,000 times. Source: RMIT.
The work is described in a recent issue of Advanced Sustainable Systems.
Meanwhile, researchers at RMIT University’s School of Science, Melbourne, Australia, have developed an ultra-efficient, low-cost catalyst for recycling used cooking oil and agricultural waste into high-value products.
Described in a recent issue of Nature Catalysis, the catalyst can make biodiesel from low-grade ingredients containing up to 50% contaminants. The researchers believe it could double the productivity of manufacturing processes for transforming waste such as food scraps — and even microplastics and old tires — into high-value chemical precursors for making medicines, fertilizers, biodegradable packaging and more.
“The quality of modern life is critically dependent on complex molecules to maintain our health and provide nutritious food, clean water and cheap energy,” says RMIT’s Adam Lee. “These molecules are currently produced through unsustainable chemical processes that pollute the atmosphere, soil and waterways. Our new catalysts can help us get the full value of resources that would ordinarily go to waste — from rancid used cooking oil to rice husks and vegetable peelings — to advance the circular economy. And by radically boosting efficiency, they could help us significantly reduce environmental pollution,” he adds.
Lee’s team synthesized a micron-scale ceramic sponge with a hierarchically porous framework in which the macropores are selectively functionalized with a sulfated zirconia solid acid coating and the mesopores with magnesium oxide solid base nanoparticles. Molecules initially enter the sponge through large pores, where they undergo a first chemical reaction (transesterification) and then pass into smaller pores where a second (condensation) reaction occurs.
It’s the first time a multi-functional catalyst has been developed that can perform several chemical reactions in sequence within a single catalyst particle; it could be a game changer for the $34-billion global catalyst market, Lee believes.
The next steps are scaling up the catalyst fabrication from grams to kilograms and adopting 3D printing technologies to accelerate commercialization.
“We’re also hoping to expand the range of chemical reactions to include light and electrical activation for cutting-edge technologies like artificial photosynthesis and fuel cells. And we’re looking to work with potential business partners to create a range of commercially available catalysts for different applications,” Lee concludes.
Seán Ottewell is Chemical Processing's editor at large. You can email him at [email protected].