Catalyst Promises Greener Styrene Production
A team of researchers at North Carolina State University (NCSU), Raleigh, N.C., has developed a catalyst that efficiently converts ethylbenzene to styrene with significantly higher yield and lower energy consumption. “Because it [styrene] is in such widespread use, we are pleased that we could develop a technology that is cost effective and will reduce the environmental impact of styrene manufacturing,” says Fanxing Li, a chemical engineering professor at NCSU. Current styrene production reportedly emits over 27 million tons of carbon dioxide.
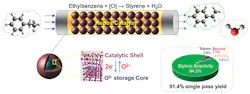
The multifunctional core-shell redox oxidative dehydrogenation (redox-ODH) catalyst acts as a heterogeneous catalyst, an oxygen separation agent, and a selective hydrogen combustion material. This converts ethylbenzene to styrene with up to 97% single-pass conversion and 94% selectivity, report the researchers. Conventional styrene production technologies have a single-pass yield of about 54%.
“We were able to prepare redox catalysts with a core-shell architecture using earth-abundant materials and a relatively simple preparation method,” notes Li. The team also determined the underlying catalytic mechanism. “This finding allowed us to fine-tune the catalyst performance by optimizing the core and shell structures and compositions to yield even better results,” he adds.
“The oxygen storage phase of the redox catalyst can be further tuned, along with improving the surface compositions, to achieve even better styrene yields by minimizing the initial less selective region under the full oxidation mode. In fact, we have developed better performing catalysts,” says Li.
Figure 1. Researchers develop a redox-oxidative dehydrogenation scheme that converts ethylbenzene to styrene using a tailored multi-functional redox catalyst. Source: Nature Communication.
The conversion process temperature is similar to traditional methods at 500–600 °C, however, the redox-ODH process requires no steam for a reaction to take place.
“In practical terms, this drastically reduces the amount of energy needed to perform the conversion,” says Yunfei Gao, a postdoctoral scholar at NCSU and co-lead author of a paper in Nature Communications on the work. The new process uses 82% less energy — and reduces carbon dioxide emissions by 79%, report the researchers.
The catalyst boasts stability and robustness, exhibiting excellent long-term performance under industrially compatible conditions, they note.
In the article, the researchers write, “These findings… provide important mechanistic insights for designing effective redox catalysts for alkylbenzene conversions.”
So far, we have tested 100 repeated redox-oxidative dehydrogenation (ODH) cycles. Longer-term validation is certainly desirable from an industrial application standpoint but it can be done through future studies,” states Li.
“Most of our experiments used high purity ethylbenzene as the feed, but we did look at a few other alkylbenzenes. The data do not suggest that the catalyst will be deactivated by common contaminants but this certainly needs to be further investigated. It would be relatively easy to test them out, elaborates Li.
The team is interested in finding an industrial partner for scaling up this technology.
“Scale up and long-term catalyst testing are the key challenges to be addressed in the next step,” says Li. “It may take 3–5 years for a pilot study.”
The team has received a patent for its work.