New Method Enables Oxygen Activation With Lanthanide Catalysts
Researchers at Rice University have demonstrated a new way to activate oxygen using lanthanide metals, a breakthrough that could expand catalyst design strategies for small-molecule transformations in chemical processing.
The study, published in the Journal of the American Chemical Society, shows how lanthanides—previously considered ineffective for this type of chemistry—can be enabled to bind and activate dioxygen through π interactions. According to the researchers, this opens the possibility of creating lanthanide-oxo species as alternatives to iron-based systems commonly used in oxidation reactions.
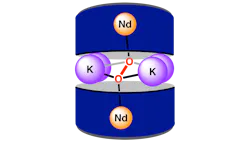
The team, led by Raúl Hernández Sánchez, developed a ligand “basket” structure that positions neodymium atoms to interact with a dioxygen molecule. This configuration creates an octacoordinate environment that facilitates previously unobserved bonding behavior, allowing the oxygen molecule to be activated and cleaved.
“We had a ligand platform that we developed a few years ago,” Hernández Sánchez said in a press statement. “You can think of it as a basket that allows us to capture metals and position them in ways to encourage specific types of bindings.”
The researchers found that under specific conditions, neodymium could engage in π interactions with dioxygen, enabling formation of highly reactive lanthanide-oxo compounds. These species could potentially be used to drive oxidation reactions or generate value-added chemicals, according to the study.
The work focused on neodymium, but the researchers said the approach may extend to other lanthanides and possibly actinides using the same ligand framework. Future research will explore the reactivity of these compounds and evaluate their potential as functional replacements for iron-oxo systems in catalytic applications.