Should You Ditch Batch Production?
Switching to continuous production of fine chemicals and pharmaceuticals now made via batch operations promises significant advantages such as faster processing, better energy efficiency and smaller-size equipment. However, realistically assessing whether such a conversion makes sense can be difficult. Now, a team led by researchers at the Institute of Chemical and Engineering Sciences (ICES) at A*STAR, Singapore, claim to have developed a practical method to comprehensively evaluate existing batch processes to enable faster yet soundly based decisions about whether to convert to a continuous process.
The goal was not to optimize a given process from first principles but rather to efficiently identify a suitable process that meets business needs, explain the researchers.
“Our methodology makes understanding the process much clearer, especially to the chemists and engineers dealing with the synthesis,” notes Soo Khean Teoh of ICES. “It makes it easier to decide to proceed or kill the idea, minimizing wasted effort.”
“The biggest challenge is that there is no ‘one-size-fits-all’ solution for changing from batch to continuous methods, because of the complex and varied chemistries involved,” comments Teoh. “We had to devise a method which guides users to understand the process in question, brainstorm about potential benefits, and help them come up with swift decisions.”
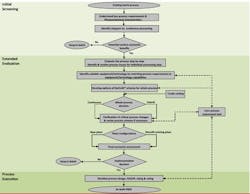
The methodology consists of three stages: initial screening, extended evaluation and process execution (Figure 1). The team already has applied the approach to three reactions with reasonably complex molecules.
Figure 1. The approach reportedly gives insights quickly on the feasibility of converting a batch process into a continuous one. Source: A*STAR.
Initial screening focuses on understanding process requirements such as capacity needed, quality constraints, where the costs of the current process arise, and business drivers for change. This involves simple assessments (e.g., yes/no/maybe) of the physicochemical characteristics and behavior of the batch process to get a rough gauge of whether continuous processing potentially offers benefits. It also calls for a parallel evaluation of possible drawbacks of continuous processing as well as process issues that differ from those of batch operations. Such a screening can lead to a quick and justifiable decision on whether or not to consider a switch, say the researchers.
Continuous processes showing promise then undergo an extended evaluation, and those found worth pursuing enter the final, project execution stage. More details on the methodology appear in a recent article in Organic Process Research & Development.
“We are working on the underpinning business case as well as some ideas in modular plant design to make the delivery of continuous easier,” says Teoh. “We expect to publish our business case thinking by the end of this year. We already have talked about modularity at conferences, but to have a complete approach to [a] modular plant will take a few years.”
“We have discussed our approach with Britest [a not-for profit company based in Daresbury, U.K., that includes pharmaceutical and fine-chemical makers as members].