Membrane Eases Flue-Gas Carbon Dioxide Removal
A new membrane separates and concentrates carbon dioxide from any type of flue gas, including gas mixtures where CO2 concentration is very low, say its developers at SINTEF, a research organization in Trondheim, Norway.
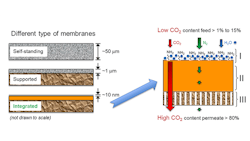
The researchers use so-called “hybrid integrated membrane technology” in which ultraviolet light integrates a nanometer-thick polymer layer onto a base polymer membrane (Figure 2). Then, a CO2-philic grafted-chain surface layer chemically functionalizes this high-permeability thin film.
The hair-like strands that rise from the surface layer contain reactive groups such as amines that bond with CO22 in the presence of water vapor. Pressure in the flue gas or a vacuum on the other side forces gas with CO2 removed across the membrane.
A 20-cm2 membrane was tested at 1.2–5 bar and from 25–55°C, and produced CO2 selectivity up to 150-fold higher than the unmodified membrane.
“The lab-scale membrane was subjected to over a thousand hours of stability testing using a variety of gas pressures, temperatures and water vapor contents. It remained intact and performed well,” explains SINTEF researcher Marius Sandru.
The next stage is gradually upscaling the membrane, initially to 500 cm2.
“We plan testing stability using real flue gas first in static tests — i.e., exposure — followed by pilot testing in natural-gas-fired and Waste2Energy pilot facilities in an ongoing project,” he adds.
Another benefit of this novel membrane — whose development also involved the chemical engineering departments at North Carolina State University, Raleigh, N.C., and the Norwegian University of Science and Technology, Trondheim — is that the base membrane is made of easily available and inexpensive olydimethylsiloxane and another polymer similar to polytetrafluoroethylene.
The surface polymerization process is not very complicated either, although Sandru admits it does require careful attention to get the best results. So, the price of this technology never will exceed that of already existing membranes, he says.
The technology suits more than treating flue gases; it could be tweaked to remove CO2 from other mixtures of interest, too —for example from hydrogen as part of hydrogen purification and from biogas mixtures.
“It is possible to graft other polymers on the surface to ‘attract’ other gases, too, but this would require knowledge of their thermodynamic compatibility. The chemical affinity of CO2 in amines is well-known,” he concludes.