High purity water and the steam produced from it constitute the lifeblood of most process plants. Equipment failures and curtailed production due to water/steam issues can cost a site hundreds of thousands of dollars or more annually. Much worse, some failures can cause injury or death. So, here, we’ll examine several of the most important issues related to proper water treatment and chemistry control in steam generators.
Let’s begin with a case history. A number of years ago, a colleague and I visited an organic chemicals plant in the Midwest that every two years or so had to replace the steam superheater bundles in four 550-psig package boilers due to internal scaling. We first were shown a recently removed bundle; roughly ¼-in.-thick deposits covered the internal tube surfaces. We then inspected the boilers and immediately noticed foam issuing from the saturated steam sample lines. Subsequent investigation revealed that total organic carbon (TOC) levels in the condensate return to the boilers sometimes reached 200 ppm — ASME guidelines [1] call for a maximum TOC concentration of 0.5 ppm in boilers of this pressure. So, it was easy to see why much foam existed in the boiler water and why impurities carried over to the superheaters on a continual basis.
[javascriptSnippet ]
The Impact Of Impurities
Impurities cause corrosion, scaling and other problems. These become more severe as boiler pressures and temperatures increase. Fortunately, the power industry has learned some lessons that directly apply to chemical plants, particularly ones that generate high-pressure steam for process needs or electrical generation. For example, Tables 1 and 2 summarize guidelines developed by the Electric Power Research Institute (EPRI) for makeup water effluent and condensate pump discharge (CPD) for heat recovery steam generators (HRSGs) [2].
Table 1. Conductivity and sodium measurements can warn of contamination ingress to the steam generator.
Table 2. Organics can break down into small-chain organic acids that impact fluid chemistry.
An examination of the effects of some of these impurities reveals why the limits are so low. Consider chlorides. Even small amounts that enter the steam generator, say, from a condenser tube leak or contaminated condensate return, if chronic and not neutralized by the boiler-water treatment program, will concentrate under deposits on boiler internals. Chloride salts in the high temperature boiler environment can react with water per:
MgCl2 + 2H2O + heat → Mg(OH)2↓ + 2HCl (1)
The hydrochloric acid produced may cause general corrosion by itself — worse yet, the acid will accumulate under deposits, where it can react with iron to generate hydrogen. The hydrogen gas molecules penetrate into the metal wall where they then combine with carbon atoms in the steel to generate methane (CH4):
2H2 + Fe3C → 3Fe + CH4↑ (2)
Formation of the gaseous methane and hydrogen molecules causes cracking in the steel, greatly weakening its strength (Figure 1). Hydrogen damage is very troublesome because it’s not easily detected. After such damage has occurred, the plant staff may replace tubes only to find that other tubes continue to rupture. I once was part of a team that had to deal with hydrogen damage in a 1,250-psig utility boiler. Operations personnel insisted on running the unit for several weeks with a known condenser leak. Even though the team did its best to maintain adequate boiler water chemistry, the ultimate outcome was extensive hydrogen damage that required a complete boiler retubing.
Figure 1. Hydrogen gas molecules penetrate into the metal wall — notice the thick-lipped failure here. Source: ChemTreat.
Measurements of conductivity and sodium are very straightforward and are excellent for detecting contaminant ingress to the steam generator. Of course, such monitoring only has real value if it leads to prompt remedial actions by chemists or operators [3]. Organic compounds, as already noted, can cause problems in the steam generator and can break down at high temperatures to form small-chain organic acids and carbon dioxide, which may significantly influence steam and condensate return chemistry.
Meeting the makeup water guidelines requires a reliable high-purity water treatment system. A very common approach employs two-pass reverse osmosis (RO) with either polishing mixed-bed ion exchange units or electrodeionization for final conditioning. Because RO membranes are very susceptible to particulate fouling, upstream filtration is required — with micro- or ultrafiltration increasingly popular for this purpose [4].
Chemical Treatment Issues
A couple of decades ago, the common belief was that all oxygen should be eliminated from boiler feedwater, as otherwise it would cause severe corrosion. This often is true when a unit is offline and air can enter the system. However, during normal operation, this idea has been proven false unless the condensate/feedwater system contains copper alloys. Regardless, this belief led to a chemical program for feedwater conditioning that became known as all-volatile treatment reducing, AVT(R), with ammonia or amine feed to establish a mildly basic pH and reducing agent (oxygen scavenger) injection to eliminate any oxygen that escaped a mechanical deaerator. For high-pressure units, the common reducing agent once was hydrazine but safer chemicals now have supplanted it.
It now is known that AVT(R) chemistry will induce flow-accelerated corrosion (FAC) in feedwater systems; this can cause wall thinning (Figure 2) and ultimately lead to catastrophic failures. In the last 30 years, several FAC-induced failures in the U.S. have resulted in fatalities.
Fig. 2. Single-phase FAC caused the substantial wall thinning shown. Source: ChemTreat.
In brief, when steam generators go into service, the carbon steel develops a thin layer of magnetite, Fe3O4. The combination of a flow disturbance and the reducing environment causes iron ions to leach out of the steel/magnetite matrix, resulting in the wall thinning. Temperature and pH affect the extent of the dissolution; it usually reaches a peak at a temperature of about 150°C and increases with lower pH (e.g., 9 and below).
So, the areas most prone to this attack are the feedwater/economizer system of conventional steam generators and the low-pressure and, sometimes, intermediate-pressure economizers and evaporators in HRSGs.
For units that contain no copper alloys in the feedwater system — which almost always is the case for HRSGs — the recommended feedwater treatment has become all-volatile treatment oxidizing, AVT(O). This program allows the small amount of oxygen that (normally) leaks in through a condenser to remain, with perhaps even a bit of supplemental oxygen injected, such that the feedwater dissolved oxygen levels stay in a 5–10 ppb range. Addition of ammonia or an amine maintains the pH in a mid- to upper-9 range. Under these conditions, the magnetite layer becomes interspersed and overlaid with a layer of ferric oxide hydrate (FeOOH) that, with elimination of the reducing environment, is very protective. However, this program only is effective in high purity water with a cation conductivity of less than 0.2 µS/cm. Otherwise, oxygen corrosion would result. So, plants where condensate return could produce elevated conductivity shouldn’t use AVT(O).
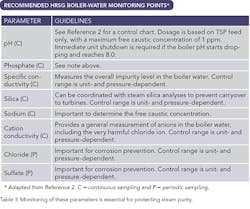
Table 3. Monitorin of these parameters is essential for protecting steam purity
Regarding feedwater chemistry monitoring, the normal limits cited in Table 2 for cation conductivity, pH and sodium for CPD apply. This is understandable because many modern industrial steam generators and virtually all HRSGs don’t have feedwater heaters; thus, the condensate chemistry should change very little in its passage to the steam generator. However, the suggested dissolved oxygen range for HRSG feedwater is 5–10 ppb. Also recommended is total iron monitoring, preferably with a corrosion product sampler, to ensure the program, whether AVT(O) or an alternative, is adequately protecting the condensate and feedwater piping. With proper chemistry, the total iron content in the feedwater should remain less than 2 ppb. If, for some reason, AVT(R) is necessary, a corrosion product sampler also will collect copper corrosion products, which provide critical data on copper corrosion control.
Boiler Water Treatment
For eighty years, steam-generation chemists have utilized sodium phosphate compounds for corrosion control and prevention of solids deposition in the waterwall circuits of drum-type boilers. Today, for high-pressure units, tri-sodium phosphate (TSP — Na3PO4), is the only recommended species, perhaps with a small amount of supplemental caustic (NaOH) for pH elevation at startups. TSP generates mild alkalinity in the boiler via :
Na3PO4 + H2O ⇔ Na2HPO4 + NaOH (3)
The alkalinity to some extent will mitigate the effects outlined in Eq. 2. TSP also provides benefit by reacting with hardness ions (calcium and magnesium) to form a soft sludge that can be blown down.
A drawback to TSP is that its solubility greatly decreases as temperatures exceed 300°F. Thus, in high-pressure units at full load, most of the phosphate precipitates on waterwall tubes and other internals. This phenomenon commonly is known as “hideout.” Many plant chemists now operate units with a bulk water phosphate concentration of ≈1–2 ppm, knowing that most of the original phosphate dose has hidden out and will redissolve at reduced boiler load or shutdown.
Boiler-water chemical treatment and monitoring in large measure is designed to protect steam purity. This is particularly true for plants that generate electricity via steam turbines. Table 3 summarizes the most important measurements.
To a large extent, the chemistry guidelines for feedwater and boiler water chemistry aim to prevent excess impurity carryover to the steam — this is especially critical if the steam drives a turbine or turbines. Steam turbines are precision machines that require careful installation, balancing and operation. (For more on steam turbines, see: “Count on Steam Turbines” .) Table 4 details the most crucial guidelines.
Table 4. Monitoring of these parameters is especially important if the steam drives a turbine.
Prevent Problems
Proper steam generation chemistry is critically important, as is the need to monitor and control chemistry at all times. Neglect of condensate return, boiler feedwater, boiler water or steam chemistry can prove quite costly both from a monetary and a safety standpoint.
In addition, proper steam generator shutdown, layup and startup procedures are critical issues, particularly to prevent offline oxygen corrosion [6,7].
BRAD BUECKER is a process specialist for Kiewit Engineering & Design, Lenexa, Kan. E-mail him at [email protected].
REFERENCES
1. “Consensus on Operating Practices for the Control of Feedwater and Boiler Water Chemistry in Modern Boilers,” ASME, New York City (1994).
2. “Comprehensive Cycle Chemistry Guidelines for Combined-Cycle/Heat Recovery Steam Generators (HRSGs),” Publ. No. 3002001381, EPRI, Palo Alto, Calif. (2013).
3. Buecker, B., and D. McGee, “An Intelligent System for Improved Water/Steam Chemistry Control and Plant Reliability,” Power Engineering (May 2014).
4. Buecker, B., “Micro- or Ultrafiltration and Reverse Osmosis: A Popular Combination for Industrial Water Treatment,” Industrial WaterWorld (January/February 2014).
5. “Technical Guidance Document: Steam Purity for Turbine Operation,” Intl. Assn. for the Properties of Water and Steam, London (2013).
6. Mathews, J., “Layup Practices for Fossil Plants,” Power (February 2013).
7. Buecker, B. and D. Dixon, “Combined-Cycle HRSG Shutdown, Layup, and Startup Chemistry Control,” Power Engineering (August 2012).