This Month’s Puzzler
Our makeup-air heat exchangers seem to have suffered ruptured tubes. On the shell side, we use 45-psig steam reduced from 200-psi boilers. We send 40% propylene glycol/water through the tubes; at the steam control valve inlet with a 50% load it’s 42 psig. The exchangers are horizontal U-tubes with ¾-in. × 0.049-in. (16 BWG) copper tubes. The tubes were rolled into a 316L stainless steel tube sheet and sealed with fluorocarbon gaskets. Both exchangers are 24-in. diameter and 6-ft long. The exchangers are about 50% oversized sometimes while running about 105% during the winter months. We run one at a time. Another concern is that the tube-side relief valve was sized using the old 77% rule — is that okay? I think a classic case of water hammer causes the crushing of a number of tubes at the top of the tube bundle that we see. When we operate at only 50%, there’s a thermal reservoir in the shell that pulls a vacuum; another engineer believes that’s the culprit. What do you think?
Check A Variety Of Items
Although data is sparse, there are several items you could look into.
Temperature gradient. Is the steam superheated or at saturation? Steam at 42 psig has a saturation temperature of ~289°F. Since you did not mention what the inlet temperature of the glycol-water mixture is, if you are using a heating media too hot and the temperature gradient (difference) is high, this may be causing additional stress on the tubes and causing premature failure. I suggest measuring the temperature difference between the glycol and steam and having a mechanical engineer determine if there is too much stress on the tubes, especially at the rolled connection at the tube sheet. Dissimilar metals — copper tubes and stainless tube sheet — could also be a concern when it comes to high temperature differences between the fluids. Check the mean metal temperature to be sure you have not exceeded the vendor’s recommendations.
If the steam is superheated, the thermal expansion stresses are more significant. You also mention you have significant over surface availability in the exchanger. Condensing steam has a high heat transfer coefficient, ~ 1,500 Btu/hr-ft2-°F. I would assume that the tube-side coefficient is controlling the overall heat transfer coefficient. You may want to consider using a different heating fluid that balances the resistances better. Is there a waste heat stream available that could be used instead of the steam? This could also help minimize stresses induced due to high temperature gradients.
The hammering may be a function of rapid condensation due to the excess surface area: condensate builds up in the shell too close to the inlet steam source.
Tube supports/baffles/tube thickness. If the tubes are not properly supported, large temperature gradients could cause failures along the outer surface of the tubes, especially at the weakest points, which may be the rolled connections. Have someone experienced in mechanical exchanger design count the tube supports and check baffle count and spacing to be sure they are adequate for the service. If you decide to change the heating medium, the baffle count and spacing may need to change to account for heat transfer and the potential resulting vibration. Your tubes are thin. Have you considered a thicker tube wall? Not knowing your flow, could a thicker tube provide the protection you need? Velocity and pressure drop are the keys to determining if you could afford a thicker tube.
Outlet glycol temperature. What is the outlet glycol/water temperature? How is it being controlled? Make sure you have not exceeded the boiling point temperature of the glycol mixture. (Propylene glycol, 40 wt-%, boils at ~219–220°F at 1 atm pressure.) You may want to check this because the steam condensing temperature is above the atmospheric boiling point of the glycol mixture. Vaporizing in the tubes will account for additional stresses that most likely will not have been addressed in the original design due to assuming no phase change on the glycol side.
Thermal reservoir. Are you condensing steam at 42 psig or higher? I’m not sure why you are pulling a vacuum if you are condensing at 42 psig. What is the chest pressure in the exchanger? If you are pulling a vacuum and the exchanger is not designed for vacuum or you do not have a vacuum breaker on the exchanger, this could be creating a problem. Perhaps you don’t need that much energy/heat transfer into the glycol stream.
Tube-side relief valve. I’m not familiar with the 77% rule. If the relief valve is not relieving, I’m not sure what part it will play in tube failure. You may want to check to see if it is leaking by. I’m not sure how much would have to be leaking by to cause a problem.
Propylene glycol degradation products. This may be a remote possibility, but propylene glycol, in the presence of O2 and metal, e.g., copper, can degrade at high temperatures into lactic acid. If the steam pressure is higher than the glycol pressure, un-deareated condensate might be entering the glycol mixture, degrading the glycol into lactic acid? I would check to be sure that you are not leaking condensate into the glycol mixture.
Eric M. Roy, principal engineer
Westlake Chemical, Houston
Add a Small Exchanger
There are several problems with this application: 1) poor selection of heat exchangers; 2) potential corrosion between the copper tubes and stainless steel tube sheet; 3) using the 77% rule for bypassing consideration of tube rupture in relief calculations.
Operating a heat exchanger at 50% of rate means that it is oversized. You don’t need a spare exchanger — you need a smaller one to meet demand when it is low. Operating a heat exchanger at a low rate affects the heat transfer coefficient: h50% ~ 0.57×hdesign based on Dittus-Boelter.
Although copper is about four times more conductive than stainless steel, it is more susceptible to rupture because it has a quarter of the allowable strength of type 316 stainless. Assuming 200-psig steam, I estimate a temperature of about 333°F based on superheat tables: http://goo.gl/DZ8flA. At this temperature, copper has an allowable stress of about 4,300 psi compared to 16,800 psi for type 316 stainless (CASTI’s guidebook on ASME B31.3: http://goo.gl/ZAORy8). One table I found showed a maximum allowable pressure for ¾-in. tubing of 470 psi. So, based on a pressure of 55 psig for the glycol (based on the 77% rule), the copper should be fine. Although copper will work-harden, strength won’t improve much. And then there is corrosion. Copper and steel, even stainless steel, although isolated by a nonconductive gasket will cause corrosion because the propylene glycol/water is conductive: the stainless steel is the anode, the copper is the cathode — the anode dissolves. Copper exposed to steam condensate always poses a corrosion concern. It might be possible for the condensate, and the dissolved CO2, always present in it, to corrode one side of the tubesheet while the water causes corrosion on the tubeside. If the steel in the tubesheet is corroding, one of the signs will be the presence of H2 in the glycol, assuming that the glycol is a trapped, recirculating liquid.
Now, let’s consider the 77% rule. It’s a dumb rule: you should always check for tube rupture in relief calculations. Besides, given the wide variation in flow rate through the exchanger, it seems doubtful that the rule applies, even in its modern form of 10/13. According to API-RP-521, section 5.19.2, tube rupture is not a concern if the low pressure is within 77% of the high pressure: this is based on the temperature-corrected hydrostatic test pressure.
There is some confusion about which temperature to use, so to be conservative, use the maximum operating temperature for the hot fluid. Note the ⅔ rule applies to older vessels; in July 2011, ASME changed the over-pressure requirements for vessels from 150% of maximum allowable working pressure (MAWP) to 130%. Regardless of ASME, you should always look at tube rupture because it might reveal design errors in the exchanger.
To get a better idea of the root cause of the tube failure, check: 1) the type of temperature control valve employed; 2) the condition of the glycol; 3) the steam supply; and 4) whether a vacuum breaker is used.
Typically, globe valves, with their slowly rising stems, are better at steam control than ball valves. The latter have been responsible for numerous water hammer problems. You should replace ball valves or add snubbers to their air supply so they close slowly. Also, check the tuning of the control valves, especially at low flow rates where valves tend to hunt for steady state.
It seems possible that plugging in the tubes may be a contributing factor to the water hammer. To see if that’s the case, take a sample of your glycol system. If there isn’t a strainer and filter, get one.
If the steam supply pressure dips, the temperature control valve goes to 100% and condensate literally will drop out of the shell with a potential for causing water hammer. You should review the isometrics for the system, model it and decide if you need loops around heavy users on the circuit to maintain a relatively constant pressure in the exchanger shells.
Finally, if you don’t have vacuum breakers on the shells get them. Make sure nothing isolates them from the shells and there are procedures and valves to vent the air that entered the shells. Trapped air can reduce heat transfer and cause hot spots that could damage tubing.
Dirk Willard, consultant
Wooster, Ohio
August’s Puzzler
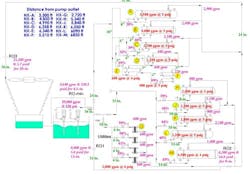
We would like to increase the cooling water flow to a tower condenser (heat exchanger A in Figure 1) from 2,900 gpm to the exchanger nameplate rating of 4,500 gpm. A review of the files shows the heat exchangers and control valves have plenty of capacity. In fact, we are concerned that so many of our heat exchangers are oversized. We’re also looking at ways to reduce the recirculation flow and pressure losses caused by the balancing orifices — we may be able to re-use the pump if we can eliminate some of this flow and the pressure losses caused by the balancing orifices. Do you think we can increase heat exchanger A to its nominal flow rate? What will this entail? Will this affect the best operating point of the pump? Do we really need the balancing orifices?
Figure 1. Plant wants to increase flow to top heat exchanger (A) to rated flow.
Send us your comments, suggestions or solutions for this question by July 10, 2015. We’ll include as many of them as possible in the August 2015 issue and all on ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Rd., Suite 400N, Schaumburg, IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you’d like to pose to our readers, send it along and we’ll be pleased to consider it for publication.