This Month’s Puzzler
We use a bank of continuously stirred reactors to raise the pH of an inorganic slurry in preparation for drying and packaging. The proper pH is critical for controlling dryer corrosion, which would result in rust contamination. Some dryer chemicals are pH-sensitive and the pH of the product must be between 6.7 and 7.9. We add finishing chemicals for packaging in the last tank. The process is fraught with problems. The pH probes in the beginning tanks foul often with different campaigns of product. Sometimes the chemicals added in the last tank result in product rejects in packaging. How can we improve this process and reduce product rejections?
Tackle Two Issues
In a broad sense, you may divide the problems into two major, but related, areas: pH sensing and pH control. Consider the following:
1. The figure shows solid caustic addition through a hopper. Excessive solids may be fouling the pH probe and giving faulty pH indication. You may consider: a) relocating the probe so that it is in an area where stream velocity is high (say, 3 to 10 ft/sec); b) providing in-situ cleaning; c) buying new probes that are more resistant to fouling/scaling; or d) trying to avoid adding caustic with lumps.
2. Make sure, if you have temperature variations in feed or reactor, to provide temperature compensation for the pH measurements. pH is sensitive to temperature.
3. pH control problems also may be due to sticky valves. Observe the motion of the valve stem — it should be smooth. Another cause of sticky valves could be the actuator. Make sure the air supply to actuator/positioner is dry and oil free, steady, and pressure is adequate.
4. If pH indications continue to show many spikes, you should check for corroded terminals, loose connections, ground-loop problems or poor wiring practices, e.g., susceptibility to electromagnetic interference (EMI). Shields and twisted pair practices are common and are effective in avoiding EMI.
5. If the pH probes are aging, consider installing new probes. Old probes are prone to offset, drift problems. They can also “freeze.” Provide periodic calibrations per manufacturer’s recommended procedures.
6. As far as control is concerned, unless the solution is buffered, a pH range of 6.7 to 7.9 could be in a steep or sensitive region where the process gain is high and a small addition of caustic could cause a big change in pH. Caustic is a strong base and is prone to cause large pH changes. Depending on the pH range you’re trying to control, for different campaigns you may have to consider multiple control valves with different Cv values to cover different pH ranges. pH, as we know, is logarithmic and not linear. Wide excursions of pH may require you to tune the loop frequently. There are some rules-of-thumb values in literature you might try. For proper control and tuning, you should create a pH and caustic addition procedure for your specific solutions.
7. Depending on the extent and severity of problems, you might consider other control schemes such as cascade or feed-forward. “pH Control Solutions” by Greg McMillan (ISA) is an excellent reference for pH control alternatives: http://goo.gl/6dWQPK. A vendor’s technical folks may also be of help.
G. C. Shah, senior HSE advisor
Wood Group Mustang, Houston
Agitation Is Important
There are two classes of problems with this process: agitation and process control. It is a small wonder the product quality control is not worse.
Let’s start with the agitation: 1) the axial flow impellers shown maximize flow but provide very little shear; 2) adding the caustic at the top of the tanks isn’t as effective in promoting dissolving as injecting it into the zone around the impeller; 3) introducing solid caustic directly into a tank is difficult because you can’t maintain a steady flow — better to dose it as a solution; and 4) even better is adding the caustic into the pumped discharge or into a mixing chamber inside each tank.
Increasing the impeller shear to liquid also increases the shear damage on the impeller. Even the current axial impellers are probably showing wear. A ceramic coating could reduce wear: check the Rockwell hardness of the coating versus the slurry. Shear is useless if it can’t get at the clumps in the liquid. Two approaches should eliminate large clumps: sifting before adding to the tank or using a high-shear mixer. Automatic sifters are available. A high-shear mixer can either be mounted offset from the tank centerline to swipe at the liquid as it swirls past or installed near the bottom of the agitator shaft with an axial impeller feeding it. This latter solution could get pricy.
The weaker the solution used to control pH, the easier it is to control because more liquid is flowing. Unfortunately, this adds to the water that must be removed by the dryer so it’s an economic tradeoff: heat cost versus quality losses from failure to achieve the target pH. There are other ways to reduce quality losses.
It may be a better idea to move the pH probes away from the beginning of the process. Why not double-up on the pH probes in the last three tanks. Another suggestion is to install the pH probes in a pumped discharge line. This will reduce scale buildup.
One final thought. Don’t expect an instrument in one tank to agree with or even be comparable with one in another tank. This is especially true for analytical instruments but generally true for all instruments. In this example, the best approach is to take a measurement with a single calibrated pH probe of samples from each of the tanks. This can be used not only to approximate the true difference between the tanks but also as a check against the probes themselves. In addition, the longer it takes for a pH probe reading to stabilize, the more the probe element has been corrupted or contaminated.
Dirk Willard, consultant
Wooster, Ohio
July’s Puzzler
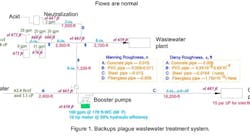
We’re having problems with our plant wastewater system (Figure 1). First, we can’t seem to get the nominal flow out of our new fiberglass neutralization system — we constantly back up in the neutralizer. Second, the drains in the plant are backing up. Our plant manager is convinced we’re seeing overflow from a 4-in. storm-water line that was in use before the process and storm-water treatment systems were separated back in the 1970s. In addition, the city water plant complains that our wastewater is spilling over into the storm water and corroding the line from two sewer sump pumps. What do you think the problem is and what can we do to address these issues?
Figure 1. Backups plague wastewater treatment system.
Send us your comments, suggestions or solutions for this question by June 12, 2015. We’ll include as many of them as possible in the July 2015 issue and all on ChemicalProcessing.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 1501 E. Woodfield Rd., Suite 400N, Schaumburg, IL 60173. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you'd like to pose to our readers, send it along and we'll be pleased to consider it for publication.