A recent ranking of the 500 greenest companies in America by Newsweek (www.newsweek.com/green) put Eastman Chemical Co. at 95, making the Kingsport, Tenn., firm one of the chemical companies cited highest.
"Unlike many of its peers, [Eastman Chemical Co.] has largely avoided environmental controversies. Has also done an excellent job reporting on emissions and waste and has set concrete targets and goals to reduce air, greenhouse gases, and hazardous waste emission levels," noted the judges in their citation.
"This recognition is further reinforcement of the sound practices and systems we have in place throughout the company to ensure a sustainable future for our employees, our communities and our environment," noted Theresa Lee, Eastman's senior vice president with responsibility for environmental, health, safety and security programs.
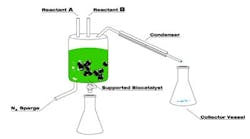
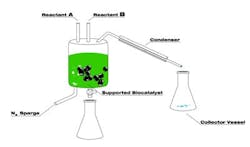
One of the reasons behind Eastman's success is its breakthrough green biocatalytic process that already has won a 2009 Presidential Green Chemistry Challenge Award.
The new process uses enzymes and closely controlled conditions to make esters, eliminating the high temperature and strong acids traditionally required.
The cosmetic and personal-care industry in North America annually consumes an estimated 50,000 tons of esters for a variety of uses including emollients, emulsifiers and specialty performance ingredients. Eastman targeted its first biocatalytic venture here because the sector tends not to follow the cyclical nature of the chemicals market and also was growing very quickly when the project started three years ago.
Commenting on driving forces behind the new process, Eastman research associate Stephanie Clendennen said, "Until recently we had a fairly stable, low-cost petrochemical supply and less emphasis on sustainability issues. This has changed so much in the last three years. Now there is a wonderful emphasis on sustainability. Along with that, technological advances such as lower-cost enzyme catalysts and membrane-based separation technologies are maturing and can be applied to more green processes."
"Generally it's all about cost. We don't deal directly with consumers much, but our customers do. And that will be where the big push forward comes from," added Eastman technology fellow Neil Boaz.
Eastman now has synthesized a variety of esters using enzymes at mild temperatures. Esterifications are driven to high conversion by removing co-product, usually water from esterification of an acid or a lower alcohol from transesterification of an ester. Filtration easily removes the immobilized enzyme, e.g., lipase. Specificity of the enzymatic conversions and relatively low reaction temperatures minimize formation of byproducts (some of which could affect color and odor), increase yield and cut energy consumption. The process reportedly can save more than 10 liters of organic solvent per kilogram of product.
"We performed an internal evaluation using published data and methods. This estimates that when this process is compared with a traditional synthetic route that uses high-temperature distillation/separation post-reaction steps, it saves up to 86% of energy usage and the same percentage of greenhouse gas emissions," noted Boaz.
"We have achieved an essentially one-step process in the laboratory (Figure 1)," said Clendennen, adding that while further commercial development depends on demand market confidence is building now. Once a decision is made, it would take about 18 months for commercial production to begin, she believes.
This growth in market confidence coincides with more and more of Eastman's major customers such as L'Oreal, Procter & Gamble and Johnson & Johnson setting their own sustainability targets.
The process is but one of a whole raft of Eastman initiatives geared to greening the company and saving money. These include: completing 107 process optimization projects, repairing 3,200 utility/steam leaks, improving the accuracy of air flow measurements on boilers and installing 82,000 feet of pipe insulation as well as recycling 1,400 tons of cardboard and paper fibers and replacing 16,841 incandescent bulbs with energy-saving fluorescent bulbs. A project to improve soap dispensers in rest rooms is saving $30,000 annually in materials and $37,000/year in labor.
A Similar Story
"Sustainability is built into the core of how we operate and is a strategic priority for us," notes Pat Loughlin, vice president of environment, health, safety and quality and chair of the sustainability council at Air Products, Allentown, Pa.
"The principles behind sustainability have always driven Air Products — for example, increasing energy efficiency and environmental problem-solving — but we haven't always called it sustainability. In the past it would have been EH&S [environmental, health and safety], or customer focus. But it's become a bigger priority for us over the last few years and is opening up new opportunities and challenges."
The company has specific targets, for example: to reduce energy consumption by 7% by 2015; to cut water use by 10% by the same year; and to decrease hazardous waste by 20% from a 2005 baseline. Air Products has trimmed toxic releases substantially over the last 10 years and now aims to maintain them at the current level.
Like Eastman, the company views process optimization as an important driver, especially for cutting energy use and generation of byproducts. Air Products also is placing more emphasis on developing uses for waste streams. For example, it now makes a saleable product out of aqueous ammonia waste via treatment in a modified distillation and purification process.
"Another example here is when we took the still bottom from one reaction and used it as a feedstock for a different product. The still bottom was produced by an amine process but now we use it as a feedstock in the manufacture of an epoxy curing agent. In terms of savings, both of these processes pay for themselves. But the primary driver is the environmental benefit," notes Loughlin.
The company is involved in a whole range of initiatives aimed at boosting the sustainability both of its own activities and those of its customers.
One is ion transport membrane (ITM) technology. At the moment, oxygen production requires large air-separation plants and cryogenic processing and so is very energy intensive. "Ceramic ITM technology has the potential to enable more economical and energy-efficient oxygen and may reduce by up to 30% the power required to produce oxygen for gasification and other energy-intensive applications, such as power, chemicals/petrochemicals and metals," says Loughlin.
An ITM project currently is underway with unnamed customers and the U.S. Department of Energy (DOE). "This is close to semi-commercial. Both the customers and the DOE are very excited about its prospects — and, of course, it fits in well with our other oxyfuel business supplying energy-intensive industries such as steel manufacturing and cement," he adds.
Air Products already has applied its oxyfuel combustion technology to increase production rates, reduce emissions and raise fuel efficiency at aluminum recyclers: producing the metal requires just 5% of the energy needed for making primary aluminum (Figure 2).
Another project involves the steam methane reforming (SMR) process that currently is responsible for almost all hydrogen production. "Not only are we looking to optimize this process, but also how to produce the gas in non-conventional ways, for example, from domestic sewage or biomass — there's a whole range of potential sources. We do have some pilot units testing these new methods but none as yet can replace SMR for commercial-scale hydrogen production in the short term. However, demand is strong and growing for hydrogen, so work on the pilots is going on."
More Process Developments
Meanwhile Bayer MaterialScience, Pittsburgh, is garnering environmental and productivity gains with its IMPACT technology. Marketed as a greener alternative to conventional polyol production processes, IMPACT has helped the company make dramatic improvements at its Channelview, Texas, plant. These include eliminating 75 million pounds of wastewater and reducing energy consumption by 80%.
The technology combines a new catalyst with an innovative process design. Continuous processing replaces a semi-batch approach that has been the industry standard for 50 years.
The catalyst is more than 10 times as reactive as other catalysts in its class and a thousand times more reactive than conventional catalysts, says Bayer. The novel process leverages a unique catalyst characteristic by which low molecular weight materials react preferentially in a mixture of molecular weights. The combination results in a process with little or no waste. The approach saves energy by eliminating the heat-up cycle of the semi-batch process and energy and waste normally associated with catalyst removal.
"The technology is being considered for implementation worldwide as plants are being upgraded or constructed, increasing the positive effects on the environment, which include energy reductions, carbon-dioxide-equivalents reductions and waste elimination," notes Jack Reese, the company's manager of polyether process development.
Dow Chemical Co., Midland, Mich., also is implementing greener technology worldwide.
"Sustainability and energy efficiency are critical initiatives at Dow," stressed Torsten Kraef, Dow Building and Construction business group vice president in early September. He was commenting on the conversion of its Varennes, Quebec, facility to the company's zero-ozone-depleting, no- volatile-organic-compound foaming agent technology for manufacturing Styrofoam extruded polystyrene foam insulation.
The Varennes' move marks the third conversion for Dow (after Hanging Rock, Ohio, and Dalton, Ga.), and its first in Canada, which, like the U.S., under the Montreal Protocol requires phase out of hydrochlorofluorocarbon 142b before January 1, 2010.
BASF, Ludwigshafen, Germany, sees significant opportunities for greener textile production. In a joint project with leading partners along the textile value chain, the company, using empirical data collected during the actual production process, has calculated the carbon footprints of specific articles. BASF textile auxiliaries and technologies then allowed the partners to cut overall CO2 emissions.
The company's after-soaping agent Cyclanon XC-W for dyeing pares processing time and water consumption compared to the conventional system. Its Color Fast Finish, an intelligent coloration system that combines dyeing, washing and finishing into one step, reduces processing time and CO2 emissions.
Safety Benefits
"Consumer safety along with environmental protection are our top priorities when coming to developing new products," states Janardhanan Ramanujalu, director and head of global business management textile chemicals.
"One outstanding example is BASF's formaldehyde-free textile processing system that ensures 'zero add-on' of formaldehyde during production. This is especially important for manufacturers of baby and children's wear, where controls for formaldehyde levels have become more stringent. Not only can customers meet today's standards, they can be rest assured also in the future," explains Ramanujalu.
Safety also is important to the sustainability efforts of Celanese Corp., Dallas, Texas. Reflecting that, its Boucherville, Quebec, plant has won a top innovation award from the Quebec Health and Safety Commission for a new automated cleaning system for a large emulsions reactor. The project took more than two years to complete and was designed to eliminate confined space entry, thus reducing the risk of potential injury from high-pressure water washing. The project also decreased the cleaning time of the reactor, thereby dramatically increasing reactor and workplace productivity.
"Safety is a cultural cornerstone and a core value of Celanese," said Jim Alder, senior vice president, operations & technical. "Our emphasis on safety is part of our commitment to society as a global chemical enterprise, as well as a means of sustainable development," he adds.
Seán Ottewell is Chemical Processing's Editor at Large. You can e-mail him at [email protected].