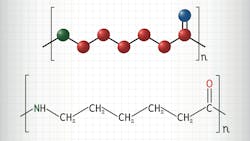
Benign, Solventless Process Depolymerizes Nylon-6
The catalyst can be reused multiple times without being deactivated.
Researchers at Northwestern University, Evanston, Illinois, have patented an organometallic catalytic-based process that depolymerizes Nylon-6 under mild, benign solvent-free conditions with what they describe as unprecedented rates and selectivity.
Based at Northwestern’s School of Engineering and its Institute for Sustainability and Engineering, the researchers used a previously developed in-house catalyst that harnesses yttrium and lanthanide ions. When this was added to melted Nylon-6 samples the plastic fell apart, reverting to its original ε-caprolactam building blocks and leaving behind no byproducts.
“This solventless process operates with catalyst loadings as low as 0.04 mol % at temperatures as low as 220°C — the mildest Nylon-6 depolymerization conditions reported to date,” write the researchers in a recent issue of Chem.
The catalyst can be reused multiple times without being deactivated.
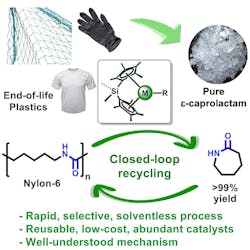
In addition to its high yield of monomers, the catalyst is highly selective and acts only on Nylon-6 polymers. As such, it could be used by industry to target Nylon-6 in large volumes of unsorted waste.
"If you don't have a catalyst that's selective, then how do you separate the nylon from the rest of waste?" asked Northwestern chemistry and engineering professor Tobin Marks, who is also the study's senior author. "You would need to hire humans to sort through all the waste to remove the nylon. That's enormously expensive and inefficient. But if the catalyst only degrades the nylon and leaves everything else behind, that's incredibly efficient," he added.
The researchers also investigated if they could create a fully circular end-of-life Nylon-6 economy by re-polymerizing recovered ε-caprolactam from a depolymerized waste fishing net. Using an anionic polymerization procedure, they obtained recycled Nylon-6 with a 99.3% yield.
Analysis revealed this Nylon-6 to be of higher quality than that of the original fishing net — prompting the researchers to point out that their new approach could both play an important part in environmental remediation and perform the first step in upcycling Nylon-6 wastes into higher-value products (Figure 1).
The Northwestern team says they have interest from potential industrial partners but did not respond to questions about the companies involved. Similarly, there was no response to inquiries as to whether a similar catalytic approach could be used to treat polymer types other than Nylon-6.
For now, the researchers are working with the Querrey InQbation Lab, Northwestern's technology accelerator facility charged with commercializing promising scientific and engineering discoveries.
About the Author
Seán Ottewell
Editor-at-Large
Seán Ottewell is a freelance editor based in Ireland. He has an impressive background in the chemical industry. After earning his degree in biochemistry at Warwick University, UK, he earned his master's in radiation biochemistry from the University of London. His first job out of school was with the UK Ministry of Agriculture, Fisheries and Food, London, where he served as scientific officer with the food science radiation unit.
From there he entered the world of publishing. In 1990, he was the assistant editor of The Chemical Engineer, later moving on to the chief editor's position. Since 1998, he has been a regular contributor to European Process Engineer, European Chemical Engineer, International Oil & Gas Engineer, European Food Scientist, EuroLAB, International Power Engineer, published by Setform Limited, London, UK.
Chemical Processing has been proud to call Ottewell Editor at Large since 2007.
He and his family run a holiday cottage in the small village of Bracklagh in East Mayo. He also fancies himself an alpaca farmer.