"Abnormal" Molecule Boasts Unusual Benefits
Metal-free N-heterocyclic carbenes (NHCs) serve an important role in catalysis. Now, a differently structured NHC promises to open up new opportunities for making chemicals and pharmaceuticals, say its developers at the University of California, Riverside, Calif.
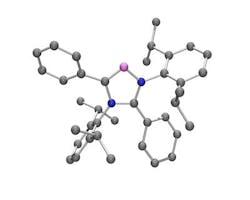
In their laboratory, they have successfully synthesized a metal-free NHC previously thought impossible to make — in which the carbene center isn't located between two nitrogen atoms as it usually is (see Figure 1). The structure of this so-called abnormal NHC or aNHC provides peculiar electronic properties that makes it among the strongest electron-donor ligands known so far, explains Guy Bertrand, a professor of chemistry at the university. "The corresponding complexes should be able to activate unreactive bonds, such as CH bonds," he notes.
The Riverside researchers challenged the assumption that aNHCs couldn't freely exist. "Many chemical species are believed to be unstable because they do not obey the rules we learned at school and, consequently, nobody tries to make them," Bertrand explains. "The aNHCs are stable at room temperature, both in the solid state and in solution, which means their application as metal-free catalysts is extremely wide, greatly benefiting industry by making possible scores of new chemical reactions."
Figure 1. Abnormal carbene carbon atom appears in violet, with nitrogen atoms in blue and normal carbon atoms in gray.
Source: University of California, Riverside.
"This study… could open new horizons and have a huge impact on the field of catalysis," notes John Schwab, who oversees organic synthesis grants at the National Institute of General Medical Sciences of the National Institutes of Health (NIH). "The potential applications to drug discovery and manufacture are exciting, since catalytic processes can help keep costs in check and be environmentally friendly, to boot."
"The next step is certainly to test metal complexes bearing aNHCs as ligands in a broad range of catalytic reactions. In parallel, we need to know which are the steric requirements that make them stable enough to be used. Moreover, other types of abnormal carbenes, with different skeletons, are being prepared," says Bertrand. NIH is funding this work.
"Our aim to is commercialize the aNHCs," says Bertrand. Industrial implementation shouldn't pose any special requirements, he believes. "Our first results show that complexes bearing aNHCs are often not sensitive to air and moisture." However, he adds: "Importantly, we need to find a better synthesis to make them available in larger quantities."
The university's Office of Technology Commercialization has applied for a patent and is hoping to line up industrial partners to develop the technology. It already is in discussions with Strem Chemicals, Newburyport, Mass.