Catalyst Combination Shines Bright
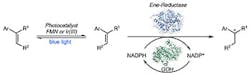
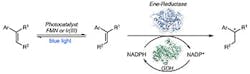
A new method combining enzymatic catalysts with photocatalysts could underpin the synthesis of a wide variety of useful compounds, believe researchers at The University of Illinois at Urbana-Champaign, Urbana, Ill. The technique brings together the reactivity of chemical catalysts with the selectivity of enzymes, they explain.
Combining enzymatic and chemical catalysts is difficult because the two types of catalysts aren’t naturally compatible. They generally function best in different media and at different temperatures, and can deactivate each other. However, the team, led by Huimin Zhao, studied several enzymes and photocatalysts and found a pair that works together.
Figure 1. Combining photocatalysts with enzymatic catalysts offers unique opportunities for synthetic chemistry, believe the researchers. Source: Huimin Zhao, University of Illinois at Urbana-Champaign.
Results showed the combined enzymatic catalysts and photocatalysts could be very productive, providing higher yields than could be achieved with enzymes alone. The researchers used the method to create a few active pharmaceutical intermediates. A recent article in Nature contains more details on their research.
Zhao foresees these two types of catalysts being used to make a variety of useful compounds. “The [compound] examples may include: (1) use of this tandem catalytic process for stereoconvergent reduction of tetra-substituted alkenes; (2) development of a similar tandem catalytic process to convert cyclic alkenes; (3) coupling of photocatalyst-catalyzed isomerization with enantioselective ene-reductase and enantioselective alcohol dehydrogenase to prepare chiral alcohols and chiral butyrolactones,” he elaborates.
The researchers aim to find photocatalysts and enzymatic catalysts combinations for more chemical reactions.
“We are exploring combinations of different photocatalysts and enzymes for additional practically useful reactions. In addition, we are investigating the possibility of using a single photocatalyst to do alkene isomerization and enzyme cofactor generation at the same time in a single system. These follow-up studies may take from a few months to a couple of years to complete.”
Both the photocatalysts and enzymes can be produced on a large scale, so the availability of catalysts won’t limit industrial applications, notes Zhao. However, some challenges remain.
“One challenge of using a tetra-substituted alkene as a substrate is that the reduction of a tetra-substituted alkene by an enzyme may result in a mixture of diastereomers. Therefore, ene-reductases that can generate a single diastereomer of product and preferentially reduce E or Z forms of tetra-substituted alkenes need to be discovered or generated by engineering existing ene-reductases. Also, photocatalyst-catalyzed isomerization and cyclization must be performed in an organic solvent that is miscible with water. Otherwise, the photocatalytic step and the enzymatic reduction step need to be performed in a two-step manner.”
The photocatalysts are quite robust, while the enzymes may have an instability issue due to the elevated temperature and decreasing pH during the reaction. However, protein engineering tools can readily improve the stability of the enzymes, notes Zhao.
Zhao’s team will pursue further research as a part of CABBI — a collaboration between the Carl R. Woese Institute for Genomic Biology and the Institute for Sustainability, Energy, and Environment along with 17 partner institutions that aims to engineer biological systems that produce non-natural compounds designed for biofuels, jet fuels, lubricants and more.
In addition, Zhao is in touch with two companies from Europe regarding potential collaboration.