Refineries: Rethink Corrosion Monitoring
Many refineries rely on equipment well past original design life. These assets, some of which now have been in operation for double that time, face an ever-increasing risk of failure due to internal corrosion attack.
Corrosion in refineries often is caused by contaminants in produced hydrocarbons that, over time, lead to deterioration of pipe and vessel walls. Loss of equipment integrity can result in unplanned downtime and costly repairs or, in the worst case, a catastrophic event posing major risk to personnel, the environment and stakeholder value.
Exacerbating the problem, many refineries no longer process the specific type of oil, such as sweet crude, they originally were designed to handle. The changing nature of oil feedstock magnifies corrosion problems in aging refineries.
Figure 1. Crudes with a high total acid number cause corrosion in areas operating above about 200°C.
For instance, in the U.S., refiners are taking advantage of the availability of light tight oils (LTOs), which afford significantly higher margins. The production of LTOs relies on the use of fracking fluids, a cocktail of chemicals for stimulating oil flow from the field. In many instances, these chemicals can end up in the crude oil feedstock to the refinery. In addition, the transportation of LTOs by railcar requires the addition of H2S passivator chemicals that can introduce other corrosion-related problems. These amine-based compounds can deposit as salts in the top section of crude towers, top pump-around and draw trays — with the resulting possibility of more corrosion.
Another example is Canadian oil sands crude, which has a high total acid number (TAN). Many of the world’s existing refineries were designed to process crudes with a TAN of 0.3 mg KOH/g or less but lots of newer crudes have a TAN of 1 mg KOH/g or more.
High TAN crudes create naphthenic acid corrosion, a particularly aggressive and often localized form characterized by the “orange peel” effect (Figure 1). While this issue primarily affects crude and vacuum distillation units, the gas, oil and residue products fed to downstream conversion and hydroprocessing units also can exhibit TAN levels that cause problems in feed-section equipment fabricated from carbon steel.
Refiners have two principal mitigation strategies against corrosion: upgrading the metallurgy of many or all the susceptible areas, often to expensive high-nickel alloys or titanium; or using chemical corrosion inhibition treatment.
Both strategies should include online corrosion monitoring at critical locations to verify the state of the metallurgy upgrade or the inhibitor distribution and effectiveness. Alternatively, online corrosion monitoring can validate that the existing mitigation strategy is performing adequately.
Assets At Risk
Refinery processing hardware prone to corrosion include sour-water strippers, crude and amine units, terminal jetties and many other assets.
Sour-water stripper tower corrosion and fouling from corrosion byproducts like iron sulfide are common operational problems compromising asset integrity. Tower and crude overhead sections are exposed to high levels of hydrogen sulfide and ammonia, and can experience excessive rates of ammonium bisulfide corrosion. High levels of cyanides from upstream units that concentrate in the overheads can compound corrosion risks (see Safeguard Sour-Water Strippers section below).
Free cyanides can be deposited in the wet gas stream, causing hydrogen blistering. Cyanides can destabilize any passivation (iron sulfide) layer, causing it to flake off as free iron sulfide, resulting in plugging and fouling.
Amine systems are subject to corrosion by both carbon dioxide and hydrogen sulfide in the vapor phase, the amine solution and the regenerator reflux — as well as from production of amine degradation products in the amine solution. In refineries specifically, amine systems suffer from corrosion by several components such as ammonia, hydrogen cyanide and organic acids not generally found in natural and synthesis gases; some of these will accumulate at various points around the refinery amine system.
Figure 2. Installing multiple ultrasonic units (orange) in areas at elevated risk of internal corrosion can help spot localized attack.
Monitoring Corrosion
Refineries can turn to two methods to measure corrosion: probes and ultrasonic sensors.
Corrosion probes, which have been in use since the 1960s, rely on an intrusive element with a sacrificial tip that sits in the process fluid. As the sacrificial tip corrodes, its electrical resistivity changes. The corrosion of the sacrificial tip is used to infer the level of corrosion being experienced by the surrounding equipment.
While simple to use, corrosion probes suffer from two disadvantages:
1. The center-line measured corrosion at the tip may not match the corrosion rate at the pipe wall.
2. The tip often corrodes away after two to three years while many refineries now operate five or more years between major turnarounds.
Ultrasonic measurement is a well-established technique for determining metal wall thickness. The technique involves the generation of ultrasound from a transducer placed directly onto the metal surface. The ultrasound travels through the metal until it is reflected off the back wall. The time difference between the sending and reflected signals correlates to the wall thickness.
Traditional ultrasonic manual inspection techniques only provide a snapshot of equipment integrity. Typically, personnel take measurements every six months to five years. Such long intervals between measurements pose a significant safety risk because a serious event can happen in a matter of hours or days. These traditional methods can’t provide the accuracy, quality and frequency of data necessary to find problems, so mitigation can’t be optimized without interrupting operations.
A Newer Option
Today, refiners instead can opt for permanently installed, wireless ultrasonic wall-thickness-monitoring sensors for corrosion monitoring. The units generate on a continuous basis the data required to make proper decisions and provide this information directly to plant personnel.
These ultrasonic sensors are non-intrusive, so their installation cost is low, and can be mounted almost anywhere. Wireless data retrieval eliminates the need for cables, further decreasing installation and ongoing operating costs. Moreover, power packs should last until the next plant turnaround (typically, nine years’ service is achievable). The simplicity of installation and long power-pack life make ultrasonic sensors well suited for use in remote locations only accessible during turnarounds.
Each sensor has a measurement footprint of approximately 1 cm2, which is similar to the area required for manual ultrasound inspection. Thus, the probability of detecting localized corrosion attack using a single sensor is small. To increase the odds of detection, sensors often are installed as multi-point arrays at high-risk locations (Figure 2).
Process temperature variations affect all ultrasound-based measurements due to the change in speed of sound through the metal. The latest generation of sensors uses an integrated thermocouple to measure the metal surface temperature and can automatically compensate the wall thickness data for temperature variations.
Wall thickness data from these sensors can go directly via the wireless network to PC-based analysis packages such as Adaptive Cross Correlation (AXC) software from Emerson (Figure 3). It can analyze and display information from dozens or even hundreds of corrosion sensors in a plant or refinery, and informs plant personnel when it discovers a problem.
Giving plant personnel access to this kind of corrosion information enables them to make the right decisions at the right time about when and where to carry out critical maintenance to support safer and more-economic operations.
Figure 3. Analysis software can detect as little as 10 microns of wall loss.
Installing the non-intrusive corrosion sensors, a wireless network and PC-based software to process the data doesn’t require a multi-day project requiring asset shutdown. Actually, deploying a real-time wireless corrosion monitoring system at strategic locations on the outside of equipment only takes a matter of hours without any interruption to refinery operations.
Improve Corrosion Monitoring
Beset with aging assets and crude feedstocks that are becoming ever-more aggressive from a corrosion standpoint, refineries face an increasing risk of equipment failure due to internal corrosion attack.
When equipped with timely information about corrosion problems, plant personnel can spot the dangers and take preventive action before corrosion presents a major operational risk. The enhanced insight provided by these real-time data allows refineries to improve safety, reduce operating costs and boost production from their aging assets. This can mean the difference between profit and loss, and between asset survival and extinction.
Safeguard Sour-Water Strippers
Sour-crude processing at refineries, unlike sweet-crude processing, often results in excessive levels of nitrogen, which leads to production of cyanides such as hydrogen cyanide. Cyanides can create corrosion issues in the sour-water system. Produced in the downstream conversion units (such as the fluid catalytic cracker or delayed coker), cyanide compounds concentrate into the water phase of the main fractionator overhead.
So, refiners can gain significant benefits from installing sensors continuously measuring wall thickness in high-risk areas of sour-water stripper towers. The monitoring data from these sensors enable determining if corrosion is taking place. This is especially valuable for understanding the correlation between corrosion rates and changes in feedstock or process conditions, minimizing the risk of leaks and fostering better forecasting of equipment retirement dates.
Continuous corrosion monitoring systems support the optimization of corrosion prevention and mitigation strategies, and also can provide data to justify decisions to upgrade to corrosion resistant alloys.
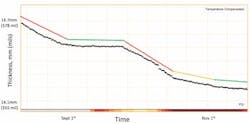
Figure 4. Monitoring led to a process change — higher reboiler duty, made permanent in November — that reduced corrosion to a tolerable level.
For example, a European refiner installed several of Emerson’s Permasense sensors around its sour-water stripper tower for general corrosion monitoring purposes. The focus was on the overhead condenser/overhead line, feed/effluent exchanger, tower bottoms line and reboiler outlet. In the course of routine monitoring, the refiner observed high corrosion at the overhead condenser outlet, which is fabricated from carbon steel.
Initially, the corrosion rate equalled 2.3 mm/y (91 mpy). However, testing on the unit showed that altering operating conditions, notably raising the reboiler duty, could significantly reduce the corrosion in this location. After operating conditions were permanently changed, corrosion fell to within tolerable limits in this area of the tower (Figure 4).
The increased reboiler duty produced more steam rising up the stripper, so more water was condensed in the overheads. This served two purposes: it reduced the hydrogen sulfide and ammonia partial pressure in the overhead system by dilution; and provided a washing of any ammonium bisulfide salts in the overhead condenser, thereby avoiding under-deposit corrosion.
Optimizing the reboiler duty extended the lifetime of the overhead exchanger and line by many years, resulting in a significant saving for the refinery from deferred equipment retirement and replacement costs. This was achieved because of the direct and rapid feedback on the impact of process condition changes on corrosion rates.
JAKE DAVIES is director of Permasense Technologies, a unit of Emerson Automation Solutions based in Horsham, U.K. E-mail him at [email protected].