THIS MONTH'S PUZZLER
Our hydrogenation system, consisting of a column filled with catalyst, an interchanger and a knockout drum under vacuum, produces excess hydrogen. The drum is meant to separate the hydrogen from the product, which is then pumped to the tank farm. The vacuum pump surges constantly, unable to find a sweet spot. The pump vents to atmosphere perhaps as much as 3 std. ft3/min of hydrogen. Our plant manager wants to know if there's a way to recover the hydrogen and stabilize the level feeding the pump. How can we address this situation? Is there any way to limp along with this process until it's convenient to shut it down?
ALTER THE CONTROL SCHEME
There are a number of issues here, so let's start with the explicit one — recovery of the hydrogen. The vacuum discharge must be connected to the hydrogen input stream, upstream of the existing H2 flow meter. The new control loop will need a buffer vessel, flow controller, flow transmitter and a compressor. The H2 flow must be totalized so that the hydrogen supply makes up the difference. Before this can be done a hazard and operability study is needed to comply with management of change.
The vacuum pump hunting comes down to poor control design. Two relatively simple improvements can be made, either individually or together. One, install a buffer vessel between the knockout drum and the vacuum pump with a pressure control valve between the knockout drum and the new buffer vessel. The second is to reduce the pumpout rate from the knockout drum. It is likely that the rate of change of pressure and the poor vacuum pump response are caused by the pump curve. A full solution would require a shutdown but the situation might be improved by altering the control parameters.
Edward Fish, director/consultant
Pandora Consulting, Manchester, England
RUN BATCH FOR NOW
I see several design flaws in this process: the vent stack, the configuration of the hydrogenation column and the controls.
My first thought is that the knockout drum is undersized. Horizontal drums are about 150% more efficient than vertical drums. The only reason for choosing the latter is to save plant floor space. Then, I noticed that the feed is co-current.
Counter-current is usually the preferred arrangement because the weak stream runs into the strong stream at either end of the equipment, resulting in the greatest driving force. Hydrogenation is very exothermic. I recall that hydrogenation of fatty acids can be as high as 25 kcal per mole. Maybe running counter-current could decrease the wasted H2 — if the temperature rise permits.
The vent stack is a different matter. I assume that the process is outside and the stack is well above any building. Most chemical companies and refiners have been forced to get rid of these in favor of flare stacks. Although H2 is lighter than air, there is always the risk of unfavorable wind conditions. H2 has a nasty habit of infiltrating enclosures. Then there is the blue, nearly invisible flame.
The most immediate concern, though, is how to keep the process running now. Try reducing the pressure and temperature of the column (gas expansion). Also, try reducing the ratio of H2 to oil. Perhaps, by watching the column temperatures, the column could be run by batch. Monitor the product quality to ensure the reaction is still going on; recycle the oil to the column feed tank until product quality meets specifications.
To recycle the H2, design a recycle line from the vent back to the hydrogen source. First, you'll need to add a second, larger knockout drum, maybe another at the source. The current one continues under vacuum but the first drum should operate with enough pressure to pump the hydrogen back to the source. The current drum should catch the oil from the new one. Trapping oil vapors may be a problem with recycle; take care in designing the drum mesh-pad mist eliminators.
Dirk Willard, contract staff engineer
Hemlock Semiconductor, Hemlock, Mich.
MARCH'S PUZZLER
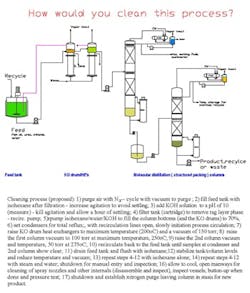
At our batch specialty chemicals plant the production manager wants us to use waste isohexane as a cleaning solvent. He proposes pumping the isohexane, which is available from a nearby process, into our columns prior to cleaning for startup. The commissioning engineer opposes using it on the grounds that a safer cleaning solvent, such as spent methanol or acetone, could be available. He suggests using methanol because it's cleaner. We're cleaning two knockout pots and two distillation columns that have been used for several months in the production of fish oil (eicosapentaenoic acid and docosahexaenoic acid). The material safety data sheet for fish oil shows the following: closed-cup flash point, 149°C; auto-ignition point, unknown; and conditions to avoid, "oxygen." The columns each contain one eight-ft. bed of structured packing. Normally, the columns operate at a maximum of 100-torr with electric thermosiphon reboilers. The condensers use chilled glycol. (See Figure 1 for proposed cleaning process.) What do you think of the commissioning engineer's objection? Is there a better approach for cleaning the process?
Send us your comments, suggestions or solutions for this question by February 11, 2011. We'll include as many of them as possible in the March 2011 issue and all on CP.com. Send visuals — a sketch is fine. E-mail us at [email protected] or mail to Process Puzzler, Chemical Processing, 555 W. Pierce Road, Suite 301, Itasca, IL 60143. Fax: (630) 467-1120. Please include your name, title, location and company affiliation in the response.
And, of course, if you have a process problem you'd like to pose to our readers, send it along and we'll be pleased to consider it for publication.