Greener Carbon Dioxide Reduction Beckons
A fully recyclable, highly selective and tunable metal-free catalyst for carbon dioxide (CO2) reduction could offer an environmentally friendly path to reducing CO2 while also yielding industrially important formate products, say researchers at the Tokyo Institute of Technology (Tokyo Tech), Tokyo, and the Renewable Energy Research Center at Japan’s National Institute of Advanced Industrial Science and Technology (AIST),Tokyo.
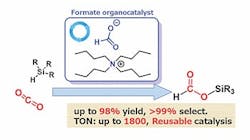
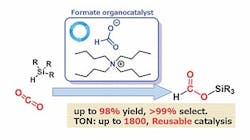
Their research focuses on hydrosilylation, a promising sustainable approach for CO2 reduction; however, current methods use catalysts containing expensive, rare metals. The Tokyo Tech/AIST team knew from a previous study that formate salts show promise as catalysts; so, by comparing how well different organic catalysts could achieve hydrosilylation of CO2, the team identified one, tetrabutylammonium (TBA) formate, that achieved 99% selectivity and produced the desired product, silyl formate, with a 98% yield. The reaction occurred rapidly (within 24 hours) and under mild conditions, at a temperature of 60°C. The catalyst also has a turnover number of up to 1,800, which exceeds previous results by more than an order of magnitude.
“Although we did expect formate salts to exhibit good catalytic activity, TBA formate showed much higher selectivity, stability and activity that went beyond our expectations,” says team leader Ken Motokura of Tokyo Tech’s Department of Chemical Science and Engineering.
In addition, the team found that using toluene as a solvent makes the catalyst reusable, and Lewis basic solvents such as N-methylpyrrolidone (NMP) and dimethyl sulfoxide (DMSO) can accelerate the reaction. An article in ACS Sustainable Chemistry & Engineering contains more detail.
The high turnover number and the catalyst’s reusability indicates a high level of robustness that Motokura suspects may stem from the rapid exchange reaction between the catalyst and silyl formate product, resulting in increased catalyst stability.
Figure 1. Formate salts serve as active and selective catalysts for the hydrosilylation of CO2. Source: Ken Motokura.
Silyl formate can be converted easily to formic acid, which can serve as an important hydrogen carrier, for example, in fuel cells. The high reactivity of silyl formate enables its conversion into intermediates for the preparation of organic compounds such as carboxylic acids, amides and alcohols.
“This efficient transformation technique of CO2 to silyl formate will expand the possibilities for CO2 utilization as a chemical feedstock,” Motokura believes.
Because TBA formate is a readily available and sustainable organic compound, Motokura doesn’t foresee any issues with producing the catalyst on a larger scale. However, one current drawback to commercial-scale reduction of CO2 is the use of expensive hydrosilane as a reducing agent.
To address this, the team is looking into silicon-based waste compounds, such as residual silane and metallic silicon from the silicone and solar panel industry. “A preliminary evaluation of new catalyst systems using such reducing agents is ongoing. We will report these reaction results within one or two years,” notes Motokura.
Increasing separability and reusability of the catalyst also is necessary, he adds. “Now we are trying to develop high-performance-supported formate catalyst for more practically useful reaction systems.”