Current methods for processing methanol operate at relatively high temperatures and pressures, require expensive catalysts, yield a range of non-specific products and typically result in the release of carbon dioxide into the atmosphere, note researchers at the University of California, Los Angeles (UCLA) Henry Samueli School of Engineering and Applied Science. They have created a more efficient way to turn methanol into useful chemicals, such as liquid fuels, while reducing carbon dioxide emissions.
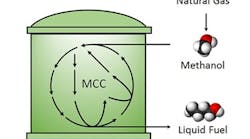
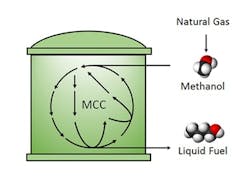
Figure 1. Researchers develop more efficient way to convert methanol to liquid fuel. Source: UCLA Engineering
“Our solution to methanol processing could be a major part of large-scale production that is much lower in cost than current standards,” says James C. Liao, chair of the UCLA Department of Chemical and Biomolecular Engineering and lead researcher on the project.
The team synthesized longer-chain molecules at room temperature and ambient pressure via a non-oxidative glycolysis pathway modified to convert methanol instead of sugar. They used a set of purified enzymes to demonstrate the biocatalytic pathway, termed the methanol condensation cycle (MCC), and were able to produce ethanol and butanol. The process releases no carbon dioxide.
“This is the first demonstration of selectively making carbon-carbon bonds from methanol only under ambient conditions. Thus, it may be cheaper and environmentally friendlier.”
In addition to higher-chain alcohols, MCC can be used to produce compounds derived from acetyl-coenzyme A, including fatty acids, alkanes, and polyketides, notes Liao.
More details appear in a recent issue of the Proceedings of the National Academy of Sciences.
Liao believes the cell-free system could be a viable application for larger-scale production after optimizing the conditions for enzyme and intermediates stability.
“Cell-free processes can, in principle, achieve 100% theoretical yield, and the production rate is linearly scalable with enzyme activity. It is not limited by the microbial metabolic activity. A cell-free process is closer to a chemical process then to a biological process, and involves less uncertainty. However, challenges include the cost of enzymes, the stability of the enzymes and the intermediates.”
Liao admits that many challenges remain. “For example, the rates are still too low. We will need to improve the activities of enzymes involved. We will also need to reduce the cost of enzymes, and increase the stability of enzymes, if a cell-free process is to be used. We will need to engineer MCC into an organism, if a microbial process is to be used.”
“We will continue to improve the performance of MCC, and then engineer MCC into a host organism. We aim to improve the rate of MCC in vitro and then implement it in one of the common host organisms. Once it is introduced in a host, it can be used to produce a variety of compounds,” he adds.
If all goes well, Liao’s team expects to conduct trials on a pilot-plant scale within 3–5 years — an “aggressive goal” he says. “However, it depends on the continued funding beyond the ARPA-e [Advanced Research Project Agency-Energy] project,” he adds. (The ARPA-e REMOTE program has funded the project since last year and will continue to for at least 6 months.)
Liao says they’ve received some inquiries from companies interested in cooperating on further development of the technology, and are selectively considering some options.