Let's say you're trying to neutralize acidic industrial waste before discharging it into the plant waste treatment system. Laboratory results don't match the values of the on-line pH analyzers (Figure 1), and you're spending more than you should on chemical reagent for neutralization. Swings of more than 3 in the pH value, possibly caused by extreme temperature changes from day to night, confound your control system. Maintenance costs are high because you're cleaning and calibrating the pH sensors constantly, and plant waste treatment personnel are complaining about your discharge stream.
COMMON CHALLENGES
One reason laboratory results don't agree with on-line analyzers' readings might be temperature. When pH is checked by taking grab samples to a laboratory or measuring them with handheld devices, frequently the temperature differs from that at the sensor sampling point.
The pH of a solution can change due to the effect of temperature on the dissociation of weak acids and bases, and especially the dissociation of water. Any solution with a pH of 7 or above will show some degree of temperature dependence. How much depends on the composition of the solution and the process temperature extremes.
The best option is to use a pH analyzer with compensation routines that account for solution pH changes due to temperature. You simply enter the temperature coefficient of the solution (pH change per °C) into the analyzer. If the solution's composition changes, however, the temperature coefficient might require adjustment.
Higher temperatures also affect the amount of reagent necessary. For example, a soap manufacturer was trying to neutralize a solution that was pH 9 at 60°C, but the pH reading would overshoot and drift compared with grab samples. At 25°C, neutralizing 1,000 liters of the waste stream required 10 ml of caustic solution. At 60°C, it took 100 ml — ten times as much.
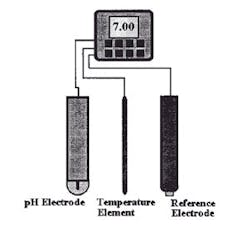
High temperatures can degrade the pH sensor itself so selection of the installation location demands care. For example, pH control is used to prevent corrosion in boilers by keeping the water slightly alkaline. Unfortunately, no pH sensor on the market today can withstand the temperatures found in typical industrial boilers. Therefore, the pH sensor is placed in a cooled and pressure-reduced side stream. In addition, monitoring highly pure boiler water requires specialized sensors.
In a beer-making process, water pushes green beer (wort) to a whirlpool to prepare for fermentation. Normally, electrical conductivity measurements determine the phase interface between the push water and the beer. However, some beer products have conductivity levels similar to that of water. In those cases, pH measurements can be used to detect the phase interface, because beer has a significantly different pH value than push water or clean-in-place (CIP) chemicals like sodium hydroxide.
The wort typically is 90°C, clean-in-place chemicals are 80–90°C, and the push and rinse water are at ambient temperature. This sharp temperature change produces thermal stress that can cause the glass electrode to crack and the sensor to fail. The sensor also must withstand the hot caustic phases, which decrease its sensitivity to pH changes. Finally, the sensor must tolerate high flow velocities.
REAGENT DOSING DIFFICULTIES
The pH scale is logarithmic, so when a solution approaches neutrality (pH = 7), a small addition of reagent can cause a large change in pH (Figure 2). The further the solution is from 7.0, the more reagent is needed to adjust pH.
If the reaction tank is large and the pH sensor is located far from the reagent dosing point, it can take a long time for the sensor to detect the change. The sensor will continue to call for reagent long after enough has been added to achieve neutralization. This leads to overshoot and oscillation, a pH value higher or lower than desired, and wasted reagent.
In a single-tank batch control application, you can use a timer to prevent overshoot by delaying the addition of reagent — thus allowing enough holdup time, mixing and chemical reaction so the sensor achieves a representative pH value. While avoiding dead time is desirable in most control loops, dead time often is a necessity in pH control.
Another common mistake is trying to adjust pH in a pipe. This usually is a bad idea because the reagent often doesn't mix properly with the process fluid. Reaction tanks with agitators are best for ensuring more complete mixing.
Reagents may be more viscous than water, and require time to mix and react. You should locate tank exits to give reagent the maximum time to react. For example, when using heavy reagents, the tank should discharge at the top instead of at the bottom.
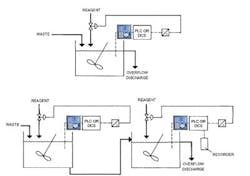
Batch control may not be practical when there's a high volume of waste, and may not work well when the holdup time in the reaction tank is less than five minutes or when wide swings occur in the pH value of the waste. Instead, consider a continuous neutralization process (Figure 3).
One reaction tank will suffice if the change required is less than 2 pH units and the amount of inflow will allow a 3–5-minute holdup time for reactions to take place. Two tanks usually are required if the change is 3–5 pH units; three tanks generally are necessary if the change is larger than 5 pH units.
DOWN AND DIRTY
The two biggest mechanical challenges facing pH sensors are temperature extremes, which can crack the glass sensor, and process fluids that coat or poison the sensor elements (Figure 4). Specific process-fluid-induced problems include:
1. High concentrations of hydroxyl ions (very high pH) can shorten the life of the electrode.
2. Hydrofluoric acid can dissolve pH glass.
3. Sulfides can poison a silver-based reference electrode.
4. Undissolved solids and liquids can coat a pH sensor.
5. Fluids can contaminate the reference solution, causing drift.
6. Certain fluids can degrade electrical connections within the sensor.
Manufacturers of pH sensors usually have solutions for all these problems. For example, to combat coating, a jet spray cleaner can be attached to the sensor to keep it clean. To avoid reference solution poisoning, its chamber can be refilled with a highly viscous reference gel using a syringe.
The sterilization required in many biopharmaceutical processes to avoid cross-batch contamination or unwanted growths can harm pH sensors. The sterilization system typically uses steam; exposure to high temperature steam and rapid thermal shock can shorten a sensor's life.
1. Readings vary with temperature.
2. The pH scale is nonlinear and logarithmic in nature.
3. High temperature cracks sensor glass.
4. Laboratory readings don't match on-line sensor values.
5. Holdup time isn't sufficient.
6. Control valve is incorrectly sized for reagent flow.
7. Sensor is fouled or damaged.
8. Mixing is inadequate.
In applications where the pH sensor must be removed for cleaning on a regular basis, or to protect it from steam during sterilization, you can install a retraction device that allows removal of the sensor without shutting the process down.
IS THE SENSOR WORKING?
Cracked glass and fouled sensors cause incorrect pH measurements and slow the response time. However, unless the sensor fails completely and sends no signal, operators can find it difficult to detect problems. So, many plants frequently pull sensors for cleaning and calibration, at a large cost in labor and process downtime, to avoid problems. Enter the modern pH sensor with built-in diagnostics.
Some instrument vendors use a digital communications protocol such as HART on the instrument's 4–20-mA output to send diagnostic information to the control system, and employ software that analyzes the sensor readings to determine if a problem exists.
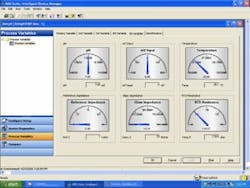
For instance, a transmitter like the Rosemount 5081-P simultaneously measures glass and reference impedance, raw sensor output, RTD resistance, pH and temperature. A glass sensor normally has an impedance of 50-150 MΩ; if cracked, impedance will drop to 0–5 Ω. A similar analysis of reference impedance can indicate sensor coating. Asset management software (Figure 5) can send alerts about glass breakage or coating buildup, and schedule appropriate maintenance.
Unnecessary maintenance can be a large expense. One pharmaceutical company found that pH sensors accounted for 46% of all field maintenance work at a batch fermentation facility. It installed sensors that send diagnostics via HART as well as asset management software, and saved over $110,000 per year in maintenance costs.
1. Pressure and temperature range of process fluid
2. Composition of process fluid
3. Reagent added to control pH
4. Required pH change
5. Process flow rate
6. Process piping layout and installation requirements
7. Availability of reaction tanks
TALK TO THE EXPERTS
While this may be your first time trying to control pH in a particular service, sensor manufacturers likely have worked on numerous similar applications and, thus, can give you useful advice. Other reliable resources can be consultants, engineering firms and system integrators well versed in pH control and measurement. Table 2 lists some of the variables crucial for selecting a sensor that suits your needs.
The expert might even warn you off using a pH sensor. For example, if you're trying to control the concentration of a caustic solution, a conductivity sensor might be a better choice.
In other applications, especially where there's a lot of acid with a pH of less than 1 or base with a pH of more than 13, using measurements made by a conductivity, a near-infrared or a refractive index analyzer may make more sense. These analyzers may provide better accuracy because they're more linear at pH extremes.
The expert might recommend using a wireless pH transmitter far downstream as a final check on pH, e.g., before a stream enters the sewer system. In many cases, the U.S. Environmental Protection Agency requires this reading weekly; the wireless device and its associated wireless network may offer savings over sending a maintenance team out to take grab samples.
The control and measurement of pH often is difficult, and most chemical plants can't afford to employ a full-time expert. The solution is to use plant personnel's deep understanding of the process and the marketplace to select the right partner, and to work together to choose the correct sensor for the particular application.
DAVE JOSEPH is Irvine, Calif.-based senior industry manager for the Rosemount Analytical Division of Emerson Process Management. E-mail him at [email protected]