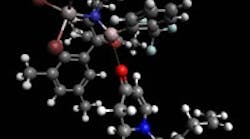
IMPACT OF LEWIS ACID
Figure 1. Lewis acid (top) shields one side of the substrate (bottom), favoring formation of the desired enantiomer. Source: Technical University of Munich.
The Lewis acid is bulky and has a specific spatial structure that shields part of the enone substrate
(Figure 1). This prompts production of a single enantiomer because the Lewis acid-complexed enone requires a lower excitation energy than the substrate alone, explain Bach and doctoral student Richard Brimioulle in a recent article in Science. "The energy is not sufficient for the non-specific reaction of the uncomplexed substrate," adds Brimioulle. The Lewis acid is released upon formation of the product and then can react with the next molecule.Currently, the researchers mainly are focusing on determining optimal Lewis acids for other substrates. "Key challenges are to find Lewis acids which combine a strong absorption shift (i.e., selective excitation of the enone in the complex) with a high enantioface differentiation," says Bach."The [2+2] photocylocaddition is the shortest and, for many products, the only method to generate the cyclobutane ring. In this respect, everyone who needs to make cyclobutanes in enantiomerically pure form should be interested in our method," he notes.The technique may suit selective synthesis of many different substances, the researchers believe. They foresee it enabling quick and efficient production of even unusually complicated molecular frameworks from simple starting materials.