Catalysts Get a New Twist
Embedding catalytic groups in foldamers, oligomers with well-defined helical structures, offers a route to high enantioselectivity, say researchers at New York University, New York City. This is because the chain-like twisted scaffold controls possible interactions, explain Kent Kirshenbaum, Michael Ward and Galia Maayan. Choice of catalyst and its location on the chain afford great flexibility to optimize performance for specific reactions.“[Our compounds] can be synthesized with an astonishing array of different chemical functionalities as oligomer side chains… This feature enables us to tune the overall physicochemical attributes of our catalysts to meet a variety of requirements,” stresses Kirshenbaum, an associate professor in the department of chemistry.One prospective application is for making drugs. The synthesis of some pharmaceuticals involves chiral molecules although only one of the mirror-image forms provides desired product. “It is crucial that a catalyst correctly makes a distinction between similar structures,” notes Kirshenbaum. “Our molecules are particularly interesting in that they are ‘selective’ — they will recognize one type of target molecule and catalyze its conversion.” More details appear in a recent paper in the Proceedings of the National Academy of Sciences.
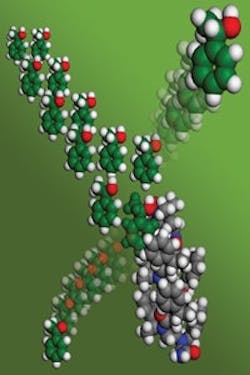
Twisted Structure
Figure 1. Chain-like molecule forms a helical scaffold that provides enantioselectivity. Source: Galia Maayan/Christopher Nannig, New York University.“These peptidomimetic compounds seem fairly bullet-proof. We know they are resistant to the actions of proteases, which is of interest for biological applications. We know that the folding and conformation is very resistant to thermal denaturation, which is promising for high temperature catalysis. And we know that these molecules can fold in a variety of solvents,” says Kirshenbaum. “Also, these oligomeric molecules are ideal for library synthesis… The ability to screen libraries of catalysts rapidly and cheaply for the desired catalytic performance is another important advantage.”“Our approach enables… known (and commercially available) achiral catalysts to be incorporated into the peptoid scaffold, which can be the same for all catalytic systems. Therefore, there is no need to develop a process or production line for each (new) specific chiral catalyst. This should make the mass production of asymmetric catalysts cheaper and technically simpler,” adds Maayan.“Our next step will be to embed different types of catalytic centers into similar helical scaffolds. We intend to demonstrate that this is a generalizable approach to selective catalysis,” notes Kirshenbaum. He hopes the work will lead to a general class of robust catalysts that exhibit enzyme-like behavior.A key challenge is to develop an understanding of how to predict which sequences will provide the desired selectivity, he says. “Once we learn the rules to connect different molecular folds to desired functions, there should be many new tricks and new tasks we can teach our molecules to perform,” he believes. “A great trick would be to perform catalysis only upon some external signal… We would like to be able to switch catalytic function on and off. A great task would be to perform catalysis within a living system.”The researchers hope to line up industrial partners to help move the development toward commercialization.