Nickel Catalyst Cuts Costs and Waste Issues
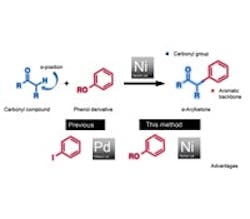
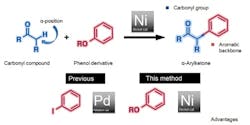
Researchers in Japan have developed a method for nickel-catalyzed cross-coupling of carbonyl compounds and phenol compounds to produce α-arylketones that are useful in pharmaceutical and organic syntheses. The method not only replaces the typical palladium catalyst with a less-expensive nickel one but also uses phenol derivatives (esters and carbamates) that usually are cheaper and more readily available than the haloarenes conventionally employed — avoiding the generation of halogen- or sulfur- containing waste, they note.
Professors Kenichiro Itami and Junichiro Yamaguchi of the Institute of Transformative Bio-Molecules at Nagoya University, Nagoya, Japan, led the team that developed the process. “Since 2009, we have been looking at the potential of nickel as a cross-coupling catalyst,” notes Yamaguchi. “Through our endeavors to develop improved conditions, we have found that phenol derivatives can act as readily available aryl coupling partners.” A key element in coming up with a successful method was the identification of an air-stable ligand, 3,4-bis(dicyclohexylphosphino)thiophene or dcypt, that improves the reactivity of nickel, leading to high yields. More details on the technique appear in a recent article in Angewandte Chemie International.
Using the nickel catalyst, the researchers have produced a variety of α-arylketone compounds, including an estrogen derivative and an amino acid tyrosine derivative. “Many natural products and bioactive compounds contain the α-arylketone framework. Our relatively low cost nickel-dcypt catalyst has exhibited high activity to couple readily available phenol derivatives in high yield,” says Itami.
Yields often are better than those achieved with palladium catalysts but the palladium system is still better in terms of robustness, Itami admits. Key challenges still to address include the development of an enantioselective catalytic system and the activation of unactivated sp3 C-H bonds.
“We want to show that this new catalyst system is applicable to even more broad class of organic substrates,” Itami adds. For instance, the researchers intend to use the method to produce amino acids and peptides that don’t occur naturally, he notes.
Already several companies have expressed interest in making the catalysts and ligands commercially available and using the reaction for the production of pharmaceuticals.
Figure 1. Using nickel catalyst and phenol derivative reduces costs and avoids generating halogen-containing waste. Source: Institute of Transformative Bio-Molecules, Nagoya Univ.