Companies Address Challenges of Batch Manufacturing
Batch manufacturing operations, such as those employed in numerous chemical processing plants around the world, are becoming increasingly complex, even as the need for flexibility, rapid response and reduced time to market grows. A session at a recent ARC Advisory Group Industry Forum highlighted the challenges facing batch manufacturing in today’s business environment and what leading manufacturers and their suppliers are doing to address these challenges.
Among other issues, the session addressed how standards, IT technology, cloud-based solutions and advanced analytics are improving operations and reducing overall support costs. Stefan Malmsten of AkzoNobel and Jeff Moore of Gallus Biopharmaceuticals presented compelling case studies. Matt Holland of GE Intelligent Platforms and Steve Schneebeli of Malisko Engineering joined them for a panel discussion that focused on how to best leverage industry standards, such as ISA-88 and ISA-95, as well as cloud and Internet of Things (IoT) technologies to create business value. (To learn more, read the Connected Plant: Get Ready for The Internet of Things and Understand The Difference Between IoT and M2M.)
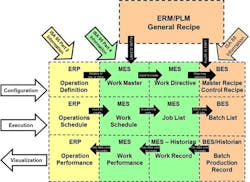
Fig. 1 Automation Suppliers Continue to Enhance Products and Services Based Upon Standards
Automation Suppliers Continue to Enhance Products and Services Based Upon Standards
ARC analyst Craig Resnick led off the session by discussing the opportunities and challenges facing batch manufacturing enterprises, as well as how automation suppliers are helping address their business needs. Resnick went on to explain how improvements in information technology and the Internet of things (IoT) drive change. This includes increased deployment of production management functions such as recipe management and plant performance monitoring and evaluation in off-site data centers.
Good Is Not Good Enough
Malmsten discussed how AkzoNobel is transforming its manufacturing operations to provide added value to its manufacturing plants and customers. The heart of this effort is to provide all the company’s plants with a single production management platform located in a remote data center to reduce both implementation and support costs, while providing a single source for analyzing production data. This eliminates the multiple, disparate and often inaccurate systems previously used to make business decisions. It also allows operators and plant engineers to benchmark their performance against colleagues within the company to make it easier to collectively identify global best practices. Malmsten further emphasized that another important aspect of the success of this project was the prescriptive use of the ISA95 standard to define the overall system architecture and the touch points with related information systems. Driving this effort forward is the company’s belief that “being good is not good enough.” Today’s competitive environment requires continual focus on AkzoNobel’s core strengths.
Engineering Design Critical to Successful ISA88 Implementations
Moore presented how Gallus Biopharmaceuticals, a leading biologics contract manufacturer, used an ISA-88-based recipe control and management system to achieve competitive advantage. The company changed its manufacturing site from a fixed large pharmaceutical facility to a contract manufacturing operation, creating the need to be more nimble and flexible to meet the needs of current and new clients. Its solution was to design product recipes, control modules, equipment, and phases based on the ISA-88 batch control standard. ISA-88 emphasizes a modular approach to batch control and the separation of the recipe procedure from the equipment procedure, allowing reusable and modifiable recipes without changing code. Some benefits achieved through a well-thought-out application-engineering design process included:
- Quicker changeover between multiple products and batch sizes;
- Shortened recipe development times;
- Faster validation times for new product recipes;
- Easier modification and creation of new recipes;
- Reduced risk of loss of product and production time;
- Lowered manufacturing costs; and
- Decreased validation costs for additional reactors by 75%.
Moore emphasized that the extra time up front on the area model and getting the operators involved as soon as possible paid big dividends for the company. Manufacturers should recognize that advances in information technology and upcoming changes as a result of IoT drive change that is transforming business processes. They should also recognize that standards play an important role in the design of process controls, and automation systems, as well as in defining the overall system architecture and the touch points with related information systems.