Rethink Reactor Temperature Control
Even minor differences between control schemes can dramatically affect unit performance. A recent project to improve control of a batch reactor clearly illustrates this.
The reactor produces small batches of a high-value specialty polymer. Reactant A is preloaded into the vessel and then Reactant B is slowly added to the batch. A mixer in the vessel provides good contacting for the first-order reaction to create the desired product. After dosing the correct amount of Reactant B, the feed is stopped and reactor contents are dumped into another vessel for further processing.
One reason for adding small amounts of Reactant B to a large quantity of Reactant A (or product) is that both Reactant A and product are much better heat sinks than Reactant B. For typical first-order kinetics, back mixing reduces the concentration and slows down temperature rise. The commonly used rule-of-thumb for most first-order reactions is that reaction rate doubles with an approximately 18°F (10°C) temperature rise.
Tight temperature control is critical to getting the right polymer properties. The process objective is to keep the reaction at as close to a constant temperature as possible. Cooling water in the reactor jacket carries away the reaction heat. The heat sink, back mixing and mixer in the vessel all aid smooth temperature control.
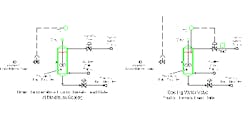
The original operation of the unit had the water supply control valve on manual. The obvious control strategy for greatest throughput is to operate the reactor at maximum cooling levels at all times (Figure 1a). Cooling water supply is set to its highest rate. The temperature controller varies Reactant B to meet the target temperature. This control scheme is simple to understand and straightforward. Simple and easy-to-understand methods tend to have high reliability and get left alone to work correctly.
However, this approach poses a problem that may not be apparent. It combines two dominant lags in series, making the system respond relatively slowly to disturbances. The first large lag is that changes in Reactant B must change the composition before reaction changes occur. The second lag is that the composition change then has to change the reaction rate before heat generation changes take place. Both of these have relatively long time constants in the system. Reactant B feed rate is small compared to the volume in the reactor. Second, the desired product forms at a relatively low temperature — hence at a relatively low reaction rate. Excursions, especially to higher-than-desired temperature, take the control system too long to correct.
[pullquote]One proposal was to use an advanced predictive controller to simultaneously change both the cooling-water and reactor rates. This would require extra instrumentation on both the cooling-water-supply pressure and temperature and a much more complicated control system. My personal experience is that most advanced control applications get turned off and abandoned sooner rather than later. Control groups simply don't have time to continuously adjust them for process and plant changes.
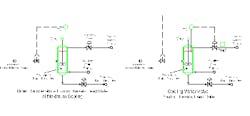
We needed a simpler alternative and came up with a solution that uses cascade single-input single-output (SISO) logic (Figure 1b). Reactor temperature controls cooling water supply. Cooling water valve position then cascades to Reactant B feed rate control. This provides much more rapid response. Position changes in the cooling water valve occur instantaneously with water flow rate. The cascade approach acts as fast as any advanced control system that attempts to simultaneously move both the Reactant-B and water-supply control valves. And, unlike advanced control methods, the logic is clear and maintenance requirements are minimal.
Choosing a reasonably high setting (90%) for the cooling water valve position gives a cooling water rate very close to the maximum possible. Reactor temperature control is stable; both reactant and utility changes are quickly dampened.
Over the past 20 years, development of really good valve positioner technology has created many opportunities to improve plant control with straightforward SISO logic. The solution here is still simple, just different from the obvious.
Andrew Sloley is a Chemical Processing Contributing Editor. You can e-mail him at [email protected].