Bio-ethanol Conversion Gets Boost
A single-pot process directly produces isobutene at high yields from ethanol made via biomass fermentation thanks to a new catalyst, say its developers at the U.S. Department of Energy's Pacific Northwest National Laboratory, Richland, Wash., and Washington State University, Pullman, Wash. "Isobutene is a versatile chemical that could expand the applications for sustainably produced bio-ethanol," notes Yong Wang, who leads the research efforts at both institutions.
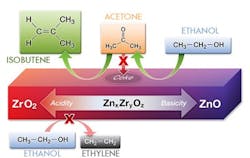
The catalyst consists of nanosized ZnxZryOz mixed oxides. It spurs ethanol dehydrogenation to acetaldehyde, its conversion to acetone, and then an acetone-to-isobutene reaction, while suppressing ethanol dehydration and acetone polymerization and coking — giving isobutene yields as high as 83%, report the researchers in a recent article in the Journal of the American Chemical Society.
"We are optimizing catalyst compositions and process conditions to further improve the yield," notes Wang. "This could take another year or so."
The catalyst requires water, so raw bio-ethanol possibly can go directly to the reactor. However, impurities in the fermentation broth might poison the catalyst and so might have to be removed. "But this shouldn't be a showstopper since purification technologies exist," he says. "Potential deactivation by coke formation is a concern. But that's not a showstopper since catalyst life is already demonstrated at >20 hours time on stream, and even without further improvement, it can be operated with a conventional swing-bed operation (one [catalyst bed] under operation and another one under regeneration to provide continuous operation)."
If everything goes as hoped for, the reaction system could reach pilot-plant stage in two-to-three years, Wang says. He doesn't envision any particular issues in producing the catalyst on a commercial scale.
Combining the catalyst with others may enable production of other chemicals such as isomers of isobutene and isooctane. "We plan to look into the possibilities of [other] one-pot reactions in a year or so," he adds.