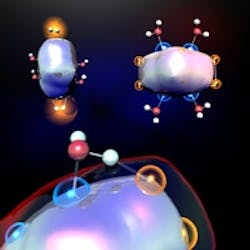
Aluminum Eases Hydrogen Generation
Exposing clusters of aluminum to water can generate hydrogen at ambient conditions without need for heat or energy, report researchers at Pennsylvania State University (PSU), University Park, Pa., and Virginia Commonwealth University (VCU), Richmond, Va.“The ability to produce hydrogen at room temperature is significant because it means that we did not use any heat or energy to trigger the reaction,” notes Shiv Khanna, a professor of physics at VCU. “Traditional techniques for splitting water to produce hydrogen generally require a lot of energy at the time the hydrogen is generated. But our method allows us to produce hydrogen without supplying heat, connecting a battery, or adding electricity. Once the aluminum clusters are synthesized, they can generate hydrogen on demand without the need to store it.”