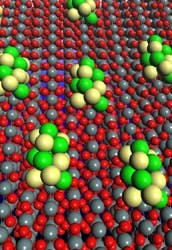
Catalyst Boosts Ethanol Fuel Cells
A new catalyst promises to enhance the prospects for ethanol-powered fuel cells, say researchers at the U. S. Department of Energy’s Brookhaven National Laboratory, Upton, N.Y., the University of Delaware, Newark, Del., and Yeshiva University, New York City. The material overcomes limitations that have hampered the use of ethanol in fuel cells.“Ethanol is one of the most ideal reactants for fuel cells,” notes Brookhaven chemist Radoslav Adzic. “It’s easy to produce, renewable, non-toxic, relatively easy to transport, and it has a high energy density. In addition, with some alterations, we could re-use the infrastructure that’s currently in place to store and distribute gasoline.” However, the fuel cells require oxidation of ethanol and current catalysts provide slow reaction speeds and form unwanted products, he adds.The electrocatalyst provides several crucial advantages, says Adzic. It oxidizes ethanol at 60°C, a good match for direct ethanol fuel cells, which typically operate at 60°C to 80°C. Moreover, it works at a positive potential of only 0.3 V while conventional catalysts require 0.7 V, which precludes their use in fuel cells. Reaction rates are significantly higher than for other catalysts. “At the mild conditions the catalyst shows around 100 times greater activity than platinum and also much greater activity than platinum-ruthenium,” he notes. And it produces mainly carbon dioxide while other catalysts largely yield acetaldehyde and acetic acid.