Fuel cells face material change
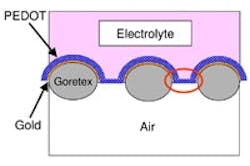
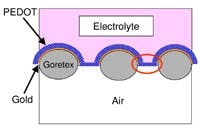
Conductive plastic deposited on breathable fabric serves both as fuel cell electrode and catalyst. Source: Monash University.
A highly conductive plastic deposited on a breathable fabric may revolutionize fuel cell design, believe researchers at Monash University, Caulfield East, Australia. The membrane functions as the air electrode, which reduces oxygen and provides performance equal to platinum-catalyzed electrodes. The technology suits metal/air batteries as well, they add.
The polymer — poly (3,4-ethylenedioxythiophene) or PEDOT — is stable and offers controllable porosity on the nanoscale. The fabric isn’t anything exotic, note the researchers — just conventional Goretex, a material that’s popular for outdoor and sports clothing, made by W. L. Gore, Newark, Del.
“The same way as waste vapor is drawn out of this material to make hikers more comfortable… so it is able to ‘breathe’ oxygen into our fuel cell and into contact with the conductive plastic,” notes Bjorn Winther-Jensen, a research fellow in the department of materials engineering.
The researchers deposited a 0.4-µ layer of PEDOT on fabric coated with a 40-nm layer of gold to further improve conductivity. They continuously tested the electrode for more than 1,500 hours, using hydrogen as the fuel source and found no signs of material degradation or performance deterioration. Oxygen conversion rates compared with those of platinum-catalyzed electrodes of the same geometry and the membrane wasn’t sensitive to carbon monoxide, which can poison platinum.
Test were conducted on one-cm2 cells from January to April this year, notes Winther-Jensen. Trials on nine-cm2 cells are underway and showing similar results. The researchers now have replaced the gold layer with stainless steel. It works just as well, Winther-Jensen says, showing that gold doesn’t contribute to the catalytic process. Other hydrophobic polymer materials besides Goretex can also be used, he adds. Monash University applied for a patent on the technology earlier this year.
The discovery ranks as probably the most important development in fuel cell technology in the past 20 years, enthuses Doug MacFarlane, a professor in the school of chemistry.
“The benefits for the motoring industry and for motorists are that the new design removes the need for platinum, which acts as the catalyst and is currently central to the manufacturing process,” he says. “The cost of the platinum component alone of current fuel cells for a small car with a 100-kW electric engine is more than the total cost of a 100-kW gasoline engine. Also current annual world production of platinum is only sufficient for about three million 100-kW vehicles, less than 1/20 of the current annual global production of vehicles.”
Replacing platinum with the PEDOT-coated material should lead to a five- to a 10-fold cut in materials and production costs for a fuel cell cathode, says Winther-Jensen.
The key challenges remaining, he notes, are: “1. Developing the active PEDOT/membrane into a three-dimensional structure — maintaining the three-phase interface (air, electrolyte and PEDOT). This is needed to meet the current densities (per cm2) required, e.g., for fuel cells in cars. 2. Engineering the cathode into cell stacks (interface with electrolyte, access of gases, etc.).”
The researchers now are looking to collaborate with specialists in these areas and also are in contact with Gore.
If all goes well, fuel cells using the technology may appear for lower-current-density applications in as little as two to three years, with automotive applications coming in six to eight years, believes Winther-Jensen. Commercial use in metal/air batteries may come even sooner, he adds.
Such conductive polymers also hold promise for industrial catalytic reactions, he says.